XENON

Overview
Xenon is a noble gas. The term noble gas is used to describe the elements in Group 18 (VIIIA) of the periodic table. The periodic table is a chart that shows how chemical elements are related to one another. "Noble gas" suggests a group of elements that is "too far above other elements" to react with them. The noble gases are also called the inert gases. That term has the same meaning. The noble gases only react with other elements under very unusual circumstances.
Xenon is very rare in the atmosphere. Its abundance is estimated to be about 0.1 parts per million. Xenon does not have many practical applications. Mostly, it is used to fill specialized lamps.
SYMBOL
Xe
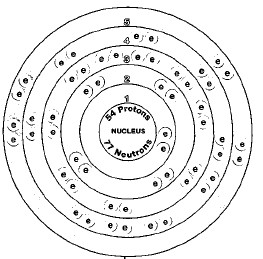
ATOMIC NUMBER
54
ATOMIC MASS
131.29
FAMILY
Group 18 (VIIIA)
Noble gas
PRONUNCIATION
ZEE-non
Discovery and naming
It took chemists more than a hundred years of careful research to understand the composition of air. In the early 1700s, they did not even understand the difference between the air around us and gases, like oxygen, carbon dioxide, and nitrogen. They used the word "air" to mean the same thing as "gas." Gases were very difficult to study. So it took a long time to figure out how various "airs" and "gases" differed from each other.
Slowly the differences became apparent. In 1774, English chemist Joseph Priestley (1733-1804) realized he could remove a separate gas—oxygen—from air. Later, other gases in the air were identified. These included nitrogen, carbon dioxide, and other noble gases. One of the last gases to be isolated was xenon.
Xenon was discovered in 1898 by Scottish chemist and physicist Sir William Ramsay (1852-1916) and English chemist Morris William Travers (1872-1961). Ramsay and Travers used liquid air to make their discovery. Here's how they did the research:
If air is cooled to a very low temperature, it changes from a gas to a liquid. As it warms up, it changes back to a gas. But this change does not take place all at once. As liquid air warms, one gas (nitrogen) boils away first. As the temperature increases further, another gas ( argon ) boils off. Still later, a third gas (oxygen) boils off.
Great care must be used in doing this experiment. The first three gases to boil away (nitrogen, oxygen, and argon) make up 99.95 percent of air. It may look as if all the air is gone after the oxygen boils away, but it isn't.
After the oxygen is gone, a tiny bit of liquid air remains. That liquid air contains other atmospheric gases. One of those gases is xenon. Ramsay and Travers first recognized the presence of xenon in liquid air on July 12, 1898. They named the element xenon for the Greek word that means "stranger."
Physical properties
Xenon is a colorless, odorless gas. It has a boiling point of -108.13°C (-162.5°F) and a melting point of C. It may seem strange to talk about the "melting point" and "boiling point" of a gas. So think about the opposite of those two terms. The opposite of melting is "turning from a liquid into a solid." The opposite of boiling is "turning from a gas into a liquid."
Thus, the boiling point of xenon is the temperature at which the gas turns into a liquid. The melting point of xenon is the temperature at which liquid xenon turns into a solid.

The density of xenon gas is 5.8971 grams per liter. That makes xenon about four times as dense as air.
Chemical properties
For many years, xenon was thought to be completely inactive. Inactive means that it does not react with any other element. Then, in 1962, English chemist Neil Bartlett (1932-) made xenon platinofluoride (XePtF 6 ). Bartlett's success inspired other chemists to try making other xenon compounds. Chemists found ways to make such xenon compounds as xenon difluoride (XeF 2 ), xenon tetrafluoride (XeF 4 ), xenon hexafluoride (XeF 6 ), xenon trioxide (XeO 3 ), and xenon oxytetrafluoride (XeOF 4 ).
In the early 1700s, they did not even understand the difference between the air around us and gases, like oxygen, carbon, like oxygen, carbon dioxide, and nitrogen. They used the word "air" to mean the same thing as "gas."
Occurrence in nature
The Earth's atmosphere contains about 0.1 part per million of xenon. Studies indicate that the atmosphere of Mars may contain about the same amount of xenon, perhaps 0.08 parts per million. The element is not known to occur in the Earth's crust.
Isotopes
Nine naturally occurring isotopes of xenon exist. They are xenon-124, xenon-126, xenon-128, xenon-129, xenon-130, xenon-131, xenon-132, xenon-134, and xenon-136. Isotopes are two or more forms of an element. Isotopes differ from each other according to their mass number. The number written to the right of the element's name is the mass number. The mass number represents the number of protons plus neutrons in the nucleus of an atom of the element. The number of protons determines the element, but the number of neutrons in the atom of any one element can vary. Each variation is an isotope.
At least 18 radioactive isotopes of xenon are known also. A radioactive isotope is one that breaks apart and gives off some form of radiation. Radioactive isotopes are produced when very small particles are fired at atoms. These particles stick in the atoms and make them radioactive.
Two radioactive isotopes of xenon—xenon-127 and xenon-133—are used in medicine. These isotopes are used to study the flow of blood through the brain and the flow of air through the lungs. In most cases, the patient inhales the radioactive gas through a mask. The xenon gas moves through the body just like oxygen or any other gas. As it travels through the body, the xenon isotope gives off radiation. The radiation can be detected by measuring devices held over the body. Doctors can tell whether the patient's lungs are working properly.
Extraction
Xenon is produced in the same way it was discovered. Liquid air is allowed to evaporate. When most other gases have boiled off, xenon is left behind. The techniques used today are much better than those used by Ramsay and Travers, of course. It is now relatively easy to capture the xenon gas in air by this method.
Uses
The primary use of xenon is in lamps. When an electric current is passed through a gas, it can give off light. Fluorescent lamps and "neon" lights are examples of this process. The kind and color of light given off depend on the gas used in the lamp. Xenon is used when a very bright, sun-like light is needed. For example, the flash units and bright lights used by photographers are often made with xenon gas.
Ultraviolet lights used to sterilize laboratory equipment may also contain xenon. The light produced is strong enough to kill bacteria. Xenon is also used in the manufacture of strobe lights. A strobe light produces a very bright, intense light in very short pulses. Strobe lights appear to "freeze" the movement of an object. Each time the light flashes on, it shines on the moving object for a fraction of a second. The object's motion can be broken down into any number of very short intervals.
Compounds
So far, xenon compounds are only laboratory curiosities. They have no practical applications. (See under "Chemical properties.")
Two radioactive isotopes of xenon are used to study the flow of blood through the brain and the flow of air through the lungs.
Health effects
Xenon is a harmless gas. Some of its compounds, however, are toxic.