SODIUM

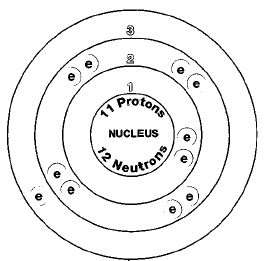
Overview
Most people have never seen sodium metal. But it is almost impossible not to see many compounds of sodium every day. Ordinary table salt, baking soda, baking powder, household lye (such as Drano), soaps and detergents, aspirin and other drugs, and countless other consumer products are sodium products.
Sodium is a member of the alkali metals family. The alkali family consists of elements in Group 1 (IA) of the periodic table. The periodic table is a chart that shows how chemical elements are related to one another. Other Group 1 (IA) elements are lithium, potassium, rubidium, cesium, and francium. The members of the alkali metals family are among the most active elements.
SYMBOL
Na
ATOMIC NUMBER
11
ATOMIC MASS
22.98977
FAMILY
Group 1 (IA)
Alkali metal
PRONUNCIATION
SO-dee-um
Compounds of sodium have been known, of course, throughout human history. But sodium metal was not prepared until 1807. The reason is that sodium attaches itself very strongly to other elements. Its compounds are very difficult to break apart. It was not until 1807 that English chemist Sir Humphry Davy (1778-1829) found a way to extract sodium from its compounds. (See sidebar on Davy in the calcium entry in Volume 1.) Sodium metal itself has relatively few uses. It reacts with other substances easily, sometimes explosively. However, many sodium compounds have many uses in industry, medicine, and everyday life.
Discovery and naming
Sodium carbonate, or soda (Na 2 CO 3 ), was probably the sodium compound best known to ancient peoples. It is the most common ore of sodium found in nature.
This explains why glass was one of the first chemical products made by humans. Glass is made by heating sodium carbonate and calcium oxide (lime) together. When the mixture cools, it forms the hard, clear, transparent material called glass. Glass was being manufactured on a large scale in Egypt as early as 1370 B.C.
The Egyptians called soda natron. Much later, the Romans used a similar name for the compound, natrium. These names explain the chemical symbol used for sodium, Na.
The name sodium probably originated from an Arabic word suda, meaning "headache." Soda was sometimes used as a cure for headaches among early peoples. The word suda also carried over into Latin to become sodanum, which also means "headache remedy."
In the early 1800s, Davy found a way to extract a number of active elements from their compounds. Sodium was one of these elements. Davy's method involved melting a compound of the active element, then passing an electric current through the molten (melted) compound. Davy used sodium hydroxide (NaOH) to make sodium.
Physical properties
Sodium is a silvery-white metal with a waxy appearance. It is soft enough to be cut with a knife. The surface is bright and shiny when first cut, but quickly becomes dull as sodium reacts with oxygen in the air. A thin film of sodium oxide (Na 2 O) forms that hides the metal itself.
Sodium's melting point is 97.82°C (208.1°F) and its boiling point is 881.4°C (1,618°F). Its density is slightly less than that of water, 0.968 grams per cubic centimeter. Sodium is a good conductor of electricity.
Chemical properties
Sodium is a very active element. It combines with oxygen at room temperature. When heated, it combines very rapidly, burning with a brilliant golden-yellow flame.
Sodium also reacts violently with water. (See accompanying sidebar.) It is so active that it is normally stored under a liquid with which it does not react. Kerosene or naphtha are liquids commonly used for this purpose.
Sodium also reacts with most other elements and with many compounds. It reacts with acids to produce hydrogen gas. It also dissolves in mercury to form a sodium amalgam. An amalgam is an alloy of mercury and at least one other metal.
Occurrence in nature
Sodium never occurs as a free element in nature. It is much too active. It always occurs as part of a compound. The most common source of sodium in the Earth is halite. Halite is nearly pure sodium chloride (NaCl). It is also called rock salt.
Halite can be found in underground deposits similar to coal mines. Those deposits were formed when ancient oceans evaporated (dried up), leaving sodium chloride behind. Earth movements eventually buried those deposits. Now they can be mined to remove the sodium chloride.
Sodium and water aren't friends
O il and vinegar don't mix. But sodium and water really don't mix! Sodium reacts violently with water. The effect is fascinating.
When sodium metal is first placed into water, it floats. But it
immediately begins to react with water, releasing hydrogen gas:
A great deal of energy is released in this reaction. It is enough to set fire to the hydrogen gas. The sodium metal reacts with water. So much heat is released that the sodium melts. It turns into a tiny ball of liquid sodium. At the same time, the sodium releases hydrogen from water. The hydrogen gas catches fire and causes the ball of sodium to go sizzling across the surface of the water.
Sodium reacts violently with water.
Sodium chloride can also be obtained from seawater and brine. Brine is similar to seawater, but it contains more dissolved salt. Removing sodium chloride from seawater or brine is easy.

Isotopes
There is only one naturally occurring isotope of sodium, sodium-23. Isotopes are two or more forms of an element. Isotopes differ from each other according to their mass number. The number written to the right of the element's name is the mass number. The mass number represents the number of protons plus neutrons in the nucleus of an atom of the element. The number of protons determines the element, but the number of neutrons in the atom of any one element can vary. Each variation is an isotope.
Six radioactive isotopes of sodium are known also. A radioactive isotope is one that breaks apart and gives off some form of radiation. Radioactive isotopes are produced when very small particles are fired at atoms. These particles stick in the atoms and make them radioactive.
Two radioactive isotopes of sodium—sodium-22 and sodium-24—are used in medicine and other applications. They can be used as tracers to follow sodium in a person's body. A tracer is a radioactive isotope whose presence in a system can easily be detected. The isotope is injected into the system at some point. Inside the system, the isotope gives off radiation. That radiation can be followed by means of detectors placed around the system.
Sodium-24 also has non-medical applications. For example, it is used to test for leaks in oil pipe lines. These pipe lines are usually buried underground. It may be difficult to tell when a pipe begins to leak. One way to locate a leak is to add some sodium-24 to the oil. If oil leaks out of the pipe, so does the sodium-24. The leaking oil may not be visible, but the leaking sodium-24 is easily detected. It is located by instruments that are designed to detect radiation.
Extraction
One way to obtain pure sodium metal is by passing an electric current
through molten (melted) sodium chloride:
This method is similar to the one used by Humphry Davy in 1808.
But there is not much demand for sodium metal. Sodium compounds are much more common. A second and similar method is used to make a compound known as sodium hydroxide (NaOH). The sodium hydroxide is then used as a starting point for making other sodium compounds.
The method for making sodium hydroxide is called the chloralkali process.
The name comes from the fact that both chlorine and an alkali metal
(sodium) are produced at the same time. In this case, an electric current
is passed through a solution of sodium chloride dissolved in water:
Three useful products are obtained from this reaction: chlorine gas (Cl 2 ), hydrogen gas (H 2 ), and sodium hydroxide (NaOH). The chlor-alkali process is one of the most important industrial processes used today.
Uses
Sodium metal has a relatively small, but important, number of uses. For example, it is sometimes used as a heat exchange medium in nuclear power plants. A heat exchange medium is a material that picks up heat in one place and carries it to another place. Water is a common heat exchange medium. Some home furnaces burn oil or gas to heat water that travels through pipes and radiators in the house. The water gives off its heat through the radiators.
Sodium does a similar job in nuclear power plants. Heat is produced by nuclear fission reactions at the core (center) of a nuclear reactor. In a nuclear fission reaction, large atoms break down to form smaller atoms. As they do so, large amounts of heat energy are given off.
Liquid sodium is sealed into pipes that surround the core of the reactor. As heat is generated, it is absorbed (taken up) by the sodium. The sodium is then forced through the pipes into a nearby room. In that room, the sodium pipes are wrapped around pipes filled with water. The heat in the sodium converts the water to steam. The steam is used to operate devices that generate electricity.
Another use of sodium metal is in producing other metals. For example,
sodium can be combined with
titanium
tetrachloride (TiCl
4
) to make titanium metal:
Sodium is also used to make artificial rubber. (Real rubber is made from the collected sap of rubber trees and is expensive.) The starting material for artificial rubber is usually a small molecule. The small molecule reacts with itself over and over again. It becomes a much larger molecule called a polymer. The polymer is the material that makes up the artificial rubber. Sodium metal is used as a catalyst in this reaction. A catalyst is a substance used to speed up or slow down a chemical reaction without undergoing any change itself.
The combination of an electric current and sodium vapor produces a yellowish glow in street lamps.
Sodium is frequently used in making light bulbs. Sodium is first converted to a vapor (gas) and injected into a glass bulb. An electric current is passed through a wire or filament in the gas-filled bulb. The electric current causes the sodium vapor to give off a yellowish glow. Many street lamps today are sodium vapor lamps. Their advantage is that they do not produce as much glare as do ordinary lights.
Compounds
Almost all sodium compounds dissolve in water. When it rains, sodium compounds dissolve and are carried into the ground. Eventually, the compounds flow into rivers and then into the oceans. The ocean is salty partly because sodium compounds have been dissolved for many centuries.
But that means that finding sodium compounds on land is somewhat unusual. They tend to be more common in desert areas because deserts experience low rainfall. So sodium compounds are less likely to be washed away. Huge beds of salt and sodium carbonate are sometimes found in desert areas.
Dozens of sodium compounds are used today in all fields. Some of the most important of these compounds are discussed below.
Sodium chloride (NaCl). The most familiar use of sodium chloride is as a flavor enhancer in food. It is best known as table salt. Large amounts of sodium chloride are also added to prepared foods, such as canned, bottled, frozen, and dried foods. One purpose of adding sodium chloride to these foods is to improve their flavors. But another purpose is to prevent them from decaying. Sodium chloride kills bacteria in foods. It has been used for hundreds of years as a food preservative. The "pickling" or "salting" of a food, for example, means the adding of salt to that food to keep it from spoiling.
This process is one reason people eat so much salt in their foods today. Most people eat a lot of prepared foods. Those prepared foods contain a lot of salt. People are often not aware of all the salt they take in when they eat such foods.
Sodium chloride is also the starting point for making other sodium compounds. In fact, this application is probably the number one use for sodium chloride.
Almost all sodium compounds dissolve in water. They tend to be more common in desert areas because deserts experience low rainfall.
Sodium carbonate (Na2CO3). Sodium carbonate is also known by other names, such as soda, soda ash, sal soda, and washing soda. It is also used as the starting point in making other sodium compounds. A growing use is in water purification
Sodium carbonate is also used to make a very large number of commercial products, such as glass, pulp and paper, soaps and detergents, and textiles.
Sodium bicarbonate (NaHCO3). When sodium bicarbonate is dissolved in water, it produces a fizzing reaction. That reaction can be used in many household situations. For example, the fizzy gas can help bread batter rise. The "rising" of the batter is caused by bubbles released when sodium bicarbonate (baking soda) is added to milk in the batter. Certain kinds of medications, such as Alka-Seltzer, also include sodium bicarbonate. The fizzing is one of the effects of taking Alka-Seltzer that helps settle the stomach. Sodium bicarbonate is also used in mouthwashes, cleaning solutions, wool and silk cleaning systems, fire extinguishers, and mold preventatives in the timber industry.
Examples of lesser known compounds are as follows:
sodium alginate (NaC 6 H 7 O 6 ): a thickening agent in ice cream and other prepared foods; manufacture of cement; coatings for paper products; water-based paints
sodium bifluoride (KHF 2 ): preservative for animal specimens; antiseptic (germ-killer); etching of glass; manufacture of tin plate
sodium diuranate, or "uranium yellow" (Na 2 U 2 O 7 ): used to produce yellowish-orange glazes for ceramics
sodium fluorosilicate (Na 2 SiF 6 ): used to make "fluoride" toothpastes that protect against cavities; insecticides and rodenticides (rat-killers); moth repellent; wood and leather preservative; manufacture of laundry soaps and "pearl-like" enamels
sodium metaborate (NaBO 2 ): herbicide
sodium paraperiodate (Na 3 H 2 IO 6 ): helps tobacco to bum more completely and cleanly; helps paper products retain strength when wet
sodium stearate (NaOOCC 17 H 35 ): keeps plastics from breaking down; waterproofing agent; additive in toothpastes and cosmetics
sodium zirconium glycolate (NaZrH 3 (H 2 COCOO) 3 ): deodorant; germicide (germ-killer); fire-retardant

Health effects
Sodium has a number of important functions in plants, humans, and animals. In humans, for example, sodium is involved in controlling the amount of fluid present in cells. An excess or lack of sodium can cause cells to gain or lose water. Either of these changes can prevent cells from carrying out their normal functions.
Dietary concerns
P eople sometimes talk about the amount of "sodium" in their diet. Or they may refer to the amount of "salt" in their diet. The two terms are similar, but not exactly alike. In the body, sodium occurs most often as sodium chloride. A common name for sodium chloride is salt.
The Committee on Dietary Allowance of the U.S. Food and Nutrition Board recommends that a person take in about 1,100 to 3,300 milligrams of sodium per day. The human body actually needs only about 500 milligrams of sodium. Studies show that the average American takes in about 2,300 to 6,900 milligrams of sodium per day.
This high level of sodium intake troubles many health experts. Too much sodium can affect the body's ability to digest fats, for example. The most serious problem, however, may be hypertension. Hypertension is another name for "high blood pressure." A person with high blood pressure may be at risk for stroke, heart attack, or other serious health problems.
Sodium is also involved in sending nerve messages to and from cells. These impulses control the way muscles move. Again, an excess or lack of sodium can result in abnormal nerve and muscle behavior. Sodium is also needed to control the digestion of foods in the stomach and intestines.
At the melt point of sodium, you also have some severe corrosion issues; so your container needs to be ceramic or a high nickel alloy (and they too will corrode). For that reason, and the chlorine gas, everything needs to be in a lab hood or some other ventilated system.
To drop the temperature, commercial manufacturers add calcium chloride to the molten salt mix to lower the melt point (freezing point depression). But despite what any literature you read says, you WILL make calcium metal. Calcium metal burns at very high temperatures (it can burn through steel), but reacts much slower than sodium with water. Commercial manufacturers will filter the sodium to remove the calcium.