Inhibitors

An inhibitor is any agent that interferes with the activity of an enzyme. Inhibitors may affect the binding of enzyme to substrate, or catalysis (via modification of the enzyme's active site), or both. Researchers use enzyme inhibitors to define metabolic pathways and to understand enzyme reaction mechanisms. Many drugs are designed as inhibitors of target enzymes. Inhibition is also a natural phenomenon. Cells regulate metabolic pathways by specific inhibition of key enzymes.
Enzyme inhibitors are divided into two classes, irreversible and reversible. Irreversible inhibition implies destruction or permanent modification of chemical groups in the enzyme. In contrast, reversible inhibitors form a complex with the enzyme that can dissociate and release the active enzyme. An enzyme E can bind either to substrate S, to form an ES complex (which can go on to products) or to inhibitor I, to form the complex EI.
There are two major types of reversible inhibitors, competitive and non-competitive. Competitive inhibition can be reversed by increasing substrate concentration, whereas noncompetitive inhibition cannot be reversed by adding more substrate.
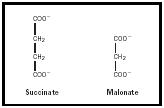
The classic example of competitive inhibition is inhibition of succinate dehydrogenase, an enzyme, by the compound malonate. Hans Krebs first elucidated the details of the citric acid cycle by adding malonate to minced pigeon muscle tissue and observing which intermediates accumulated after incubation of the mixture with various substrates. The structure of malonate is very similar to that of succinate (see Figure 1). The enzyme will bind malonate but cannot act further on it. That is, the enzyme and inhibitor form a nonproductive complex. We call this competitive inhibition, as succinate and malonate appear to compete for the same site on the enzyme. With competitive inhibition, the percent of inhibition is a function of the ratio between inhibitor and substrate, not the absolute concentration of inhibitor.
Noncompetitive inhibition is not reversed by adding more substrate. The inhibitor binds at a site on the enzyme other than the substrate binding site. The inhibitor can bind to either free enzyme, forming EI, or to the ES complex, forming EIS. Both EI and EIS are nonproductive.
Irreversible inhibitors are those that permanently disable the enzyme. The complex EI or EIS cannot dissociate, so that these are dead-end complexes. When an irreversible inhibitor is added to the enzyme-containing solution, inhibition may not be complete immediately, but increases gradually with time, as more and more enzyme molecules are modified.
Uses of Inhibitors
Among the irreversible inhibitors are organophosphorus compounds, which inhibit the enzyme acetylcholinesterase and similar enzymes. Organophosphorous compounds include nerve gases (such as sarin), that work on the human nervous system, and insecticides like malathion.
Many therapeutic drugs are enzyme inhibitors. Important examples are penicillin, which inhibits an enzyme necessary for bacterial cell wall synthesis ,

and aspirin, an inhibitor of the synthesis of molecules that mediate pain and swelling. More recent examples are drugs used in the treatment of human immunodeficiency virus (HIV) and acquired immunodeficiency syndrome (AIDS) that prevent maturation of the virus by inhibiting the HIV protease, and drugs that lower cholesterol by inhibiting a key step in cholesterol biosynthesis .
SEE ALSO Active Site ; Enzymes ; Krebs, Hans Adolf ; Neurochemistry .
Adele J. Wolfson
Bibliography
Boyer, Rodney (1999). Concepts in Biochemistry, Chapter 6 . Pacific Grove, CA: Brooks/Cole.
Bugg, Charles E.; Carson, William M.; and Montgomery, John A. (1993). "Drugs by Design." Scientific American 269(6):92–98.
Internet Resources
McMichael, Kirk. "Enzyme Inhibition and Regulation." Washington State University. Available from http://www.chem.wsu.edu/Chem102/ .