Inorganic Chemistry
The scope and boundaries of inorganic chemistry were shaped by the earlier field of organic chemistry in the mid-eighteenth and later centuries. Back in these earlier days of chemistry, all chemicals not classified as organic, in other words all chemicals, including minerals and alloys , that do not have an origin in living organisms, were placed in a very large category of "inorganic" substances. Many of the first chemists, therefore, used the describable differences between living and nonliving things to help delineate the boundaries of these two areas. Two common distinctions were that, first, organic compounds contained a unique "vital force" that arose from their biological origin and second, as a result, organic compounds could not also be synthesized or reproduced in the laboratory. Of course, both of these distinctions were later disproved, but they clearly helped form the divisions by which chemists have labeled themselves. Today a factor commonly used to distinguish between inorganic and organic substances is the presence of the element carbon. Inorganic chemistry, a vast field of study, is usually defined as the study of any compound that does not contain the element carbon, especially as it occurs in chemically bonded chains and rings that have additional chemical bonds to hydrogen atoms.
It should be noted that as chemists move increasingly toward interdisciplinary work, even this arbitrary distinction cannot adequately describe certain chemical compounds. For example, the solid material Sc 3 C 4 (three parts scandium, four parts carbon) has ethane- and propanelike organic fragments (without their hydrogen atoms) that are embedded in a solid matrix of inorganic scandium ions. In another broadly diverse field called organometallic chemistry, chains of organic-like carbon atoms form chemical bonds with inorganic metal ions such as zirconium, molybdenum, and nickel. Thus chemistry is becoming increasingly integrated and continuous, with no sharp boundary lines, as chemists realize that closely related ideas, such as chemical bonding and acid/base classifications, can be broadly applied to both complex organic substances as well as to inorganic compounds. In a recent example, an expansion of the concept of aromaticity, normally applied strictly to organic molecules such as benzene (C 6 H 6 ), was found by Lai-Sheng Wang and colleagues to explain also the enhanced stability of inorganic, metal-bonded Al 4 2− clusters.
Mono- and Polyatomic Ions
Back in the late 1800s many chemists were hard at work categorizing the known elements according to their bond valences and acid/base characteristics. However, the elements are usually not found free and pure in nature. Instead, they are usually found and worked with in the form of ions and complexes that also contain other elements. When elements combine with each other, ions tend to form, as electrons are transferred to the most electronegative elements and partially shared to form chemical bonds. An element that has gained electrons, such as O 2− , is called an anion , and one that has lost electrons, such as Al 3+ , is called a cation. In an aqueous solution , these ions will interact electrostatically with the solvent, as well as with each other. For example, ions will interact with the partially charged negative and positive regions of H 2 O, the most common solvent on Earth. Cations will be surrounded more closely by the oxygen atoms, the more negative region of the H 2 O molecules, while an anion will be more closely surrounded by the hydrogen atoms, or the more positive region of the H 2 O molecules. The product of this interaction is called a hydrated ion, shown in the following equation, and it is one of the simplest chemical species in solution (e.g., Na + , K + , F − , Cl − ):
M n+/− + H 2 O → M(H 2 O) x n+/− ( x ∼ 4–6)
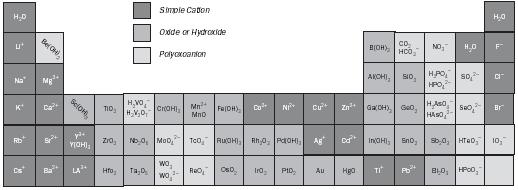
As the charges of the ions increase across the Periodic Table such as from Na + to Sc 3+ to V 5+ , or from F − to O 2− to N 3− , the bond formed between the ions and the water molecules grows strong enough to weaken and ultimately break the O–H bond. This kind of reaction with water is called hydrolysis. Products of this reaction with cations will yield hydroxides and oxides, such as Sc 2 O 3 and V 2 O 5 , and the solutions will become acidic due to the released hydrogen ions. On the other hand, the reaction with highly charged anions will tend to result in hydrides, NH 3 , and the solution will become basic due to the released hydroxide ions. Using this simple picture, which is based on the electrostatic interaction of oppositely charged ions, the principle forms of almost all the elements in nature can be qualitatively predicted (see Figure 1).
"Polyoxocations" and "-anions" are the names given to ions that exist in solution bonded to multiple oxygen atoms, such as ReO 4 − and SO 4 2− , and occurs only with the highest charged ions, such as Re 7+ and S 6+ . These high formal oxidation states prevent a large charge buildup on the ion and therefore dramatically lower the basicity of the anion and further association with H + . The sizes of many polyoxoanions may range up to the nanometer scale, with extremely large clusters, more than 100 atoms, existing in molybdenum-containing solutions. Many other types of complex ions exist in different, and less common, solvents. Among the many possible examples are fluorides, TiF 6 2− or SiF 6 2− ; cyanides, [C d(CN) 4 ] 2− ; chlorides, PtCl 6 2− ; and some organic-bonded ions, ZrCl 2 (Cp) 2 (Cp = C 5 H 5 ; cyclopentadienyl). To understand these compounds, it is first necessary to expand our acid/base definitions.
Complex Transition-Metal Ions and Compounds
Many reactions and properties that are characteristic of typical acids (H + donors) and bases (OH − donors) occur in chemical compounds that lack any hydrogen or hydroxide ions, such as in nonaqueous solvents. For example, the nitrogen group on a pyridine molecule can donate an electron pair to acidic metal cations , such as Zn 2+ or Cu 2+ , as well as to H + -containing acids such as HCl. The Zn–N, Cu–N or H–N bond formed neutralizes the acidic character of cations (H + and M n+ , discussed above), and thus the pyridine molecule functions much like a base. An expanded definition of bases is necessary to include all chemical species that can donate a pair of electrons. These are commonly called Lewis bases, after Gilbert N. Lewis, who was among the first to note this similarity in solid salts such as [C 5 H 5 NH] + Cl − or [Cd(C 5 H 5 N) 2 Cl 2 ]. Conversely, any chemical species that can accept a pair of electrons is called a Lewis acid.
The bond formed between Lewis acids and bases is termed a coordinate covalent bond , and the acid/base pair is called a coordination compound. In coordination compounds, the ligands directly bonded to the central metal atom are included within the brackets, as in [Co(NH 3 ) 6 ] 3+ , while the nonbonded ions that balance the cation charge, the counter-ions, are located outside the brackets, as in Cl − for [Co(NH 3 ) 6 ]Cl 3 . The subscripts indicate that six ammonia molecules form coordinate covalent bonds to Co 3+ . In other words, the coordination number is six. In the compound Sn(C 5 H 5 N)Cl 2 , tin is bonded to one pyridine molecule and to two chloride anions, for a total coordination number of three. The charge on Sn can be calculated by knowing the charges on the ligand species, so that if each pyridine is uncharged and the charge on each Cl is 1−, the net positive charge on Sn must be 2+ to give a neutral molecule. There are hundreds if not
thousands of possible ligands that can attach to the more than forty metal cations, so an almost unlimited number of coordination compounds may be formed. At this point, it is very useful to describe the principles that determine which metal-ligand combinations are preferred.
HSAB Classification
The question of which metal ions prefer to form complex ions with which ligands was answered partly by Ralph G. Pearson during a critical review of hundreds of naturally occurring minerals and synthetic coordination compounds. The general rule of thumb is that hard (Lewis) acids tend to combine with hard (Lewis) bases, and soft acids with soft bases. This is known as the hard-soft acid/base (HSAB) principle. The closer on the Periodic Table that a metal ion is to gold, the softest element, the softer that metal ion is, such as the soft Ag + or Cd 2+ ions. The farther from gold, the harder the metal cation, such as the hard Ca 2+ ion. For inorganic anions, the hardest is the fluoride anion. The farther away on the Periodic Table, the softer the anion, such as the soft I − anion. For complex anions, such as PO 4 3− , SO 4 2− , or CO 3 2− , the electron donor groups are the oxygen atoms, and thus these are hard anions.
This concept can explain many of the toxicological effects and the biochemistry of ions and ligands in the body. For example, soft metal cations that are soluble in the digestive tract, such as CH 3 Hg + and certain Pb salts, are extremely poisonous, as they deactivate key biological enzymes by disrupting their S–H (soft element) bonds. However, many hard metal ions, such as K + or Na + , are essential elements in the body. Examples of toxic soft bases include carbon monoxide, :CO, and the cyanide ion, :CN − , which compete with O 2 for binding to Fe 2+ in hemoglobin. Representative examples of this concept in major mineral sources include limestone, CaCO 3 (hard–hard), fluorite, CaF 2 (hard–hard), sphalerite, ZnS (soft–soft), and argentite, Ag 2 S (soft–soft).
Ligand Types
There are further properties of ligands beyond their HSAB characteristics that determine their bonding preferences to metal ions. The simplest ligands, such as pyridine or ammonia, have only a single lone pair of electrons to donate and are called monodentate ligands. However, if there are multiple unshared electron pairs, as in pyrazine or thiocyanate, these can be donated to two different metal centers to form a bridge. Alternatively, if these multiple lone pairs are situated at a distance of four to five atoms from each other, the ligand can then "wrap" itself around and donate multiple electron pairs to the same metal cation. These types of ligands are known as chelating ligands, such as in EDTA (ethylenediaminetetraacetate), which has six binding sites. Competition for binding to a metal cation will favor the chelating ligands rather than the monodentate ligands, due to the increased entropy (increased number of particles) of the products:
Co(NH 3 ) 6 3+ + EDTA 4− → Co(EDTA) 1− + 6NH 3
In both the reactants and products, cobalt retains a coordination number of six, but the number of individual chemical species increases from two to seven. In another type of ligand, a macrocycle, multiple electron donation sites are held in a rigid conformation for metal binding. This type is best illustrated by many biologically related ligands, such as chlorophyll , heme, and vitamin B 12 , which are all tetradentate macrocyclic ligands with nitrogen atom electron donors. These are used in living organisms as moving platforms with photo- and redox-active metal centers, and also as transports for certain ligands such as O 2 . Competition for binding to a metal ion between a chelating and macrocyclic ligand will favor the latter, due to entropy gains again, as the more flexible chelating ligand has greater degrees of freedom when unbound. These ligands are highly favorable architectures that transport key ions around the body.
Additional Areas
There is a great and never-ending story to the many types of complex ions, ligands, compounds, and bonding types that are known and rapidly being discovered by inorganic chemists. Some of these areas include organometallics, metal–metal bonded molecules and solids, multiply bonded main group elements, and intermetallics.
SEE ALSO Alkali Metals ; Alkaline Earth Metals ; Chalcogens ; Halogens ; Industrial Chemistry, Inorganic ; Lewis, Gilbert N. ; Semiconductors .
Paul A. Maggard Jr.
Bibliography
Li, Xi; Kuznetsov, Aleksey E.; Zhang, Hai-Feng; Boldyrev, Alexander, I.; and Wang, Lai-Sheng (2000). "Observation of All Metal Aromatic Molecules." Science 291: 859–861.
Poettgen, Rainer, and Jeitschko, Wolfgang (1991). "Scandium Carbide, Sc 3 C 4 , a Carbide with C3 Units Derived from Propadiene." Inorganic Chemistry 30(3):427–431.
Shriver, Duward, and Atkins, Peter (1999). Inorganic Chemistry . New York: W. H. Freeman and Company.
Wulfsberg, Gary (2000). Inorganic Chemistry . Sausalito, CA: University Science Books.
Comment about this article, ask questions, or add new information about this topic: