CALCIUM

Overview
Calcium is an alkaline earth metal. The alkaline earth metals make up Group 2 (IIA) of the periodic table, a chart that shows how the elements are related. They include beryllium, magnesium, strontium, barium, and radium. The alkaline earth metals are more chemically active than most metals. Only the alkali metals in Group I (IA) are more reactive.
Calcium compounds are common and abundant in the Earth's crust. Humans have used calcium compounds for hundreds of years in construction, sculpture, and roads.
Calcium metal was not prepared in a pure form until 1808 when English chemist Humphry Davy (1778-1829) passed an electric current through molten (melted) calcium chloride.
Metallic calcium has relatively few uses. However, calcium compounds are well known and widely used. They include chalk, gypsum, limestone, marble, and plaster of paris.
SYMBOL
Ca
ATOMIC NUMBER
20
ATOMIC MASS
40.08
FAMILY
Group 2 (IIA)
Alkaline earth metal
PRONUNCIATION
CAL-cee-um
Discovery and naming
It is impossible to say when humans first knew about or used compounds of calcium. Whenever they used limestone to build a structure, they were using a compound of calcium. Limestone is the common name for calcium carbonate (CaCO 3 ). Whenever humans built a statue or monument out of marble, they were using calcium carbonate in another form. Ancient Egyptians and early Greeks used mortar, a cement-like material that holds stones and bricks together. Early mortar was made by roasting or heating limestone for long periods of time. Water was then mixed with the powder, which would then dry to form a strong bond.
Humphry Davy | English chemist

H umphry Davy (1788-1829) was a major contributor to the field of electrochemistry. This is the science involving the relation of electricity to chemical changes. He is best known for discovering calcium, sodium, potassium, magnesium, strontium, and barium. He also discovered nitrous oxide and two types of lamps.
Davy grew up in Cornwall, England, in a poor family. His father, who died when Davy was a boy, had lost money in unwise investments, so Davy helped his mother pay off the debts. He disliked being a student, though he liked reading about science. In later life, he said he was happy he did not study too hard because he had more time to think on his own.
With no money for further education, the 17-year-old Davy began to work for a surgeon-pharmacist. He also started learning on his own about other subjects that interested him, such as geography, languages, and philosophy. He even wrote poems that later earned him the respect and friendship of William Wordsworth, Samuel Coleridge, and other leading English poets of his time!
At 19, Davy read a chemistry book by the famous French scientist Antoine-Laurent Lavoisier (1743-94). That book convinced him to concentrate on chemistry. For the rest of his life, Davy's career was marked by brilliant scientific explorations in chemistry and electrochemistry.
Davy discovered nitrous oxide after testing the effects of hydrogen and carbon dioxide on himself. (He liked to use himself as a human guinea pig!) Nitrous oxide is a gas consisting of nitrogen and oxygen. While studying nitrous oxide gas, he discovered that its effects often made him feel very happy or very sad. The feeling of happiness eventually gave nitrous oxide another name: laughing gas. Most importantly, though, Davy recognized that it could be used as an anesthetic. An anesthetic is a chemical used to dull pain during minor surgery.
In 1808, Davy invented the carbon arc lamp. He had proposed using carbon as the electrode material instead of metal. (Electrodes are conductors used to establish electrical contact with a nonmetallic part of a circuit.) With carbon electrodes, he made a strong electric current leap from one electrode to the other. This created an intense white light. Davy's invention marked the beginning of the era of electric light. Arc lamps are still used today.
Using his knowledge of electricity, Davy built a large battery which he used to break down substances most scientists thought were pure elements. In 1807, he discovered the element potassium. He created this by using electrolysis. Electrolysis produces chemical changes by passage of an electric current through an electrolyte. An electrolyte is a nonmetallic electric conductor. Within a week he isolated sodium in a similar way. Then in 1808, he used a slightly modified method to isolate calcium, magnesium, barium, and strontium. Davy was only 29 by the time he had discovered all of these elements!
Davy later invented the miner's lamp (now known as the Davy lamp). He learned that methane was the mine gas that caused explosions. But he realized it ignited only at high temperatures. So he designed a lamp in which the flame was surrounded by wire gauze. This reduced the heat and prevented flammable gases from igniting. This made coal-mining safer by reducing the number of explosions in mines.
Davy was rewarded by many honors and medals for his discoveries and inventions. He died of a stroke in 1829 at the age of 49.
Another calcium compound used by early civilizations was plaster of paris. Plaster of paris is made by heating gypsum, or calcium sulfate (CaSO 4 ), to remove the water that makes it crystallize. Water was added and it hardened into a brittle, cement-like substance. Until recently, it was most often used to make casts to protect broken bones. However, it has largely been replaced by fiberglass, which is lighter, yet stronger. The first mention of plaster of paris to protect broken bones can be found in a book written by Persian pharmacist Abu Mansur Muwaffaw in about 975 A.D.
By the 1700s, chemists had learned a great deal about calcium compounds. They knew that limestone, gypsum, marble, and many other commonly occurring compounds all contain a common element. They called the element calx. That word comes from the Latin term for lime. In 1807, Davy isolated the new element.
Davy invented a system for melting compounds of elements that were difficult to separate by usual methods. He passed an electric current through the compound, causing the compounds to break into parts. One of those parts was calx. He created the name calcium by adding the suffix -ium to calx; -ium is the ending used for almost all metallic elements. Davy was also able to produce free sodium, potassium, strontium, magnesium, and barium.
Physical properties
Calcium is a fairly soft metal with a shiny silver surface when first cut. The surface quickly becomes dull as calcium reacts with oxygen to form a coating of white or gray calcium oxide.
Calcium's melting point is 850°C (1,560°F) and its boiling point is 1,440°C (2,620°F). It has a density of 1.54 grams per cubic centimeter.
Chemical properties
Calcium is a moderately active element. It reacts readily with oxygen to
form calcium oxide (CaO):
Calcium reacts with the halogens—
fluorine, chlorine, bromine, iodine,
and
astatine,
The halogens are the elements that make up Group 17 (VIIA) of the
periodic table. Calcium also reacts readily with cold water, most acids,
and most nonmetals, such as
sulfur
and
phosphorus.
For example, calcium reacts with sulfur:
Occurrence in nature
Calcium is the fifth most common element in the Earth's crust. Its abundance is estimated to be about 3.64 percent. It is also the fifth most abundant element in the human body.
Calcium does not occur as a free element in nature. It is much too active and always exists as a compound. The most common calcium compound is calcium carbonate (CaCO 3 ). It occurs as aragonite, calcite, chalk, limestone, marble, and travertine, and in oyster shells and coral.
Shellfish build their shells from calcium dissolved in the water. When the animals die or are eaten, the shells sink. Over many
Isotopes
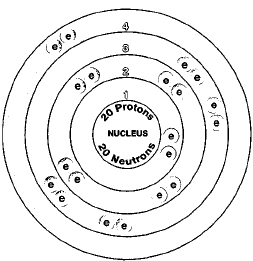
Six naturally occurring isotopes of calcium exist: calcium-40, calcium-42, calcium-43, calcium-44, calcium-46, and calcium-48. Isotopes are two or more forms of an element. Isotopes differ from each other according to their mass number. The number written to the right of the element's name is the mass number. The mass number represents the number of protons plus neutrons in the nucleus of an atom of the element. The number of protons determines the element, but the number of neutrons in the atom of any one element can vary. Each variation is an isotope.
Radioactive isotopes of calcium have also been made. A radioactive isotope is one that breaks apart and gives off some form of radiation. Radioactive isotopes are produced when very small particles are fired at atoms. These particles stick in the atoms and make them radioactive.
Two radioactive isotopes of calcium are used in research and medicine. Calcium-45 is used to study how calcium behaves in many natural processes. For example, it can be used to see how various types of soil behave with different kinds of fertilizers. The calcium-45 is used as a tracer in such studies. A tracer is a radioactive isotope whose presence in a system can easily be detected. The isotope is injected into the system at some point. Inside the system, the isotope gives off radiation. That radiation can be followed by detectors placed around the system. Calcium-45 can also be used as a tracer in the study of glassy materials, detergents, and water purification systems.
Both calcium-45 and calcium-47 can be used to study how calcium is used in the body. A doctor may think that a person's body is not using calcium properly in making bones or regulating nerve messages. The doctor can use calcium-45 or calcium-47 to find out more about this problem. The radioactive isotope is injected into the person's bloodstream. Then its path can be followed by the radiation it gives off. The doctor can then tell if the calcium is being used normally in the body.
Extraction
Pure calcium metal can be made by the same method used by Davy. An
electric current is passed through molten calcium chloride:
There is not much demand for pure calcium. Most calcium is used in the form of limestone, gypsum, or other minerals that can be mined directly from the earth.
Shellfish build their shells from calcium dissolved in the water.
Uses
Calcium metal has relatively few uses. It is sometimes used as a "getter." A getter is a substance that removes unwanted chemicals from a system. Calcium is used as a getter in the manufacture of evacuated glass bulbs. Calcium is added to the

Calcium is also used to make alloys. An alloy is made by melting and mixing two or more metals. The mixture has properties different from those of the individual metals. An alloy of calcium and cerium is used in flints found in lighters (the elements that create sparks).
Compounds
The starting point for the manufacture of most calcium compounds is limestone. Limestone occurs naturally in large amounts in many parts of the world. It is usually mined from open-pit quarries. A quarry is a large hole in the ground from which useful minerals are taken.
Limestone is first heated to obtain lime, or calcium oxide (CaO):
Lime is one of the most important chemicals in the world. It usually ranks in the top five chemicals produced in the United States. In 1996, about 19 billion kilograms (42 billion pounds or 21 million tons) of lime was produced in the United States.
Lime is used in the production of metals. It is used during the
manufacture of steel to remove unwanted sand, or silicon dioxide (SiO
2
), present in iron ore:
The product formed in this reaction, calcium silicate (CaSiO 3 ), is called slag.
Another important use of lime is in pollution control. Many factories
release harmful gases into the atmosphere through smokestacks. Lining a
smokestack with lime allows some of these gases to be captured. The lime
is known as a scrubber. Lime captures one harmful gas, sulfur dioxide (SO
2
), which is a contributor to acid rain (a form of precipitation that is
significantly more acidic than neutral water, often produced as the result
of industrial processes):
Calcium sulfite (CaSO 3 ) is a solid that can be removed from the inside of the smokestack.
"In the limelight"
At one time, lime was used as a source of light in theaters. When lime is heated to a high temperature, it gives off an intense white light. Pots of hot lime were often used to line the front of the stage. The light the pots gave off helped the audience see the performers. As a result, the performers were said to be "in the limelight." That phrase is still in use today, but lime is no longer used as a source of light in theaters.
Lime is also used in water purification and waste treatment plants. When
water combines with water, it forms slaked lime, or calcium hydroxide
(Ca(OH)
2
):
Slaked lime traps impurities present in the water as it forms. It carries the impurities with it as it sinks to the bottom of the tank.
Lime is used to make more than 150 different industrial chemicals. Some examples of these chemicals with their uses are:
Milk is a good source of calcium.
calcium alginate: thickening agent in food products such as ice cream and cheese products; synthetic fibers
calcium arsenate (Ca 3 (AsO 4 ) 2 ): insecticide
calcium carbide (CaC 2 ): used to make acetylene gas (for use in acetylene torches for welding); manufacture of plastics
calcium chloride (CaCl 2 ): ice removal and dust control of dust on dirt roads; conditioner for concrete; additive for canned tomatoes; provides body for automobile and truck tires
calcium cyclamate (Ca(C 6 H 11 NHSO 4 ) 2 ): sweetening agent (cyclamate), no longer permitted for use because of suspected cancer-causing properties
calcium gluconate (Ca(C 6 H 11 O 7 ) 2 ): food additive; vitamin pills
calcium hypochlorite (Ca(OCl) 2 ): swimming pool disinfectant; bleaching agent; deodorant; algicide and fungicide (kills algae and fungi)
calcium permanganate (Ca(MnO 4 ) 2 ): liquid rocket propellant; textile production; water sterilizing agent; dental procedures
calcium phosphate (Ca 3 (PO 4 ) 2 ): supplement for animal feed; fertilizer; commercial production of dough and yeast products; manufacture of glass; dental products
calcium phosphide (Ca 3 P 2 ): fireworks; rodenticide (kills rats); torpedoes; flares
calcium stearate (Ca(C 18 H 35 O 2 ) 2 ): manufacture of wax crayons, cements, certain kinds of plastics, and cosmetics; food additive; production of water resistant materials; production of paints
calcium tungstate (CaWO 4 ): luminous paints; fluorescent lights; X-ray studies in medicine
Health effects
Calcium is essential to both plant and animal life. In humans, it makes up about two percent of body weight. About 99 percent of the calcium in a person's body is found in bones and teeth. Milk is a good source of calcium. The body uses calcium in a compound known as hydroxyapatite (Ca 10 (PO 4 ) 6 (OH) 2 ) to make bones and teeth hard and resistant to wear.
Calcium has many other important functions in the human body. For example, it helps control the way the heart beats. An excess (too much) or deficiency (not enough) of calcium can change the rhythm of the heart and cause serious problems. Calcium also controls the function of other muscles and nerves.