Venom
It may be tempting to equate the concept of venom with poison, but to do so would be inaccurate. Many plants, for example, are poisonous but present no harm to humans because they have learned not to eat them. By contrast, one can be quite careful on a walk through the southwestern United States or Australian outback and still have an unfortunate accident involving the venom of a snake. In any given year, around 40,000 people die as a result of snakebites. To be more accurate with the definition of venom, therefore, it must be noted that venom is not just a poison, but one that is injected under the skin of the victim.
Snakes are not the only animals that use venom. Spiders, scorpions, bees, and wasps are also venomous animals. The specific venom used by these animals varies not only with its type (spider venom is different from bee venom, for example), but also within a species. Thus, some spiders are venomous but not actually a threat to adult humans, whereas the bite of other spiders is lethal unless treated with appropriate medicines.
The differences in venoms have important medical implications, but there are also similarities among many venoms. Most are rather complicated mixtures of chemicals, each of which plays some role in the action that the venom takes. Many involve some mechanism designed to immobilize the victim and are therefore targeted at nerve cells that control muscles.
Thus, snake venoms typically contain neurotoxins, but they also often include enzymes that promote various hydrolysis reactions. The neurotoxins carry out the task of immobilizing the victim by interrupting the ability

of the nerve cells to stimulate muscle movement. Hydrolysis helps make the tissues of the victim easier for the snake to digest if it is eaten. These hydrolytic enzymes may include molecules capable of breaking down collagen and phospholipids as well as other enzymes.
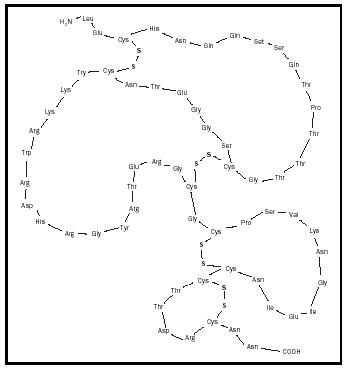
For many who study the chemistry of venoms, the neurotoxins hold particular interest. One example would be the polypeptide toxin cobrotoxin that was isolated from the Formosan cobra and analyzed in 1965 by Chen-Chung Yang, a distinguished chair professor at Tsing Hua University in Taiwan. The primary structure of this neurotoxin is indicated in Figure 1, along with some components of its secondary structure. There are sixty-two residues in the primary structure and four di-sulfide bonds in the secondary structure. If even one of these di-sulfide bonds is somehow disrupted, the polypeptide is rendered nontoxic. This points to the fact that secondary structure is important even in small polypeptides, not only full-size proteins.
The action of cobrotoxin arises from its ability to bind strongly to the receptors in postsynaptic neurons. In order for one nerve cell to signal a neighbor, it releases neurotransmitters that diffuse across a small gap called the synapse. The nerve on the other side of the synapse (the postsynaptic neuron ) has proteins that are specifically geared to sense the presence of these neurotransmitters. If these receptors are blocked, the signal is not passed on, and the nerve cell function is disrupted. Cobrotoxin is able to disrupt a specific type of receptor that is sensitive to acetylcholine . Once this polypeptide binds to the receptor, it is not released, so the nerve cell loses its ability to signal and the muscles it is supposed to trigger do not function. If the muscle that stops working is the diaphragm, the animal will not be able to breathe and it essentially suffocates. Cobrotoxin is very toxic, with an LD 50 of 65 nanograms per kilogram in mice.
SEE ALSO Globular Protein ; Neurotoxin ; Proteins ; Toxicity .
Thomas A. Holme
Bibliography
Agosta, William (2001). Thieves, Deceivers, and Killers: Tales of Chemistry in Nature.
Princeton, NJ: Princeton University Press.
Bailey, Graham S., ed. (1998). Enzymes from Snake Venom. Fort Collins, CO: Alaken.