Fullerenes
In 1985, while working in the laboratory of Richard Smalley at Rice University, graduate students Jim Heath and Sean O'Brien found that carbon aggregates in an inert atmosphere form C 60 (and to a lesser extent, C 70 ) as the most abundant species. Previous work in the Smalley laboratory had involved clusters of atoms such as silicon, germanium, and gallium arsenide. The primary motive was to find out how elements like silicon contrive to minimize their "dangling bonds" on the surface of a small, nanoscopic bare cluster composed of only 10 to 100 atoms. Dangling bonds are ones found on the edges of the cluster that do not have the full complement of electrons that would be provided by bonding to other atoms. These workers had found that some silicon clusters adopt particularly stable structures, but never become so inert that they will not readily react with another silicon atom.
In the case of carbon, the specific cluster C 60 was clearly behaving as if it had absolutely no dangling bonds as other carbon clusters continued to grow to even larger sizes in the condensing carbon vapor. Somehow the cluster had arranged in geometrical form to eliminate all dangling bonds. The only reasonable structure was a spherical one—a soccer ball—in which each carbon atom had the full complement of electrons. This epic-making work was published in the scientific journal Nature in 1985; senior investigators Richard E. Smalley, Robert F. Curl, and Harold W. Kroto were awarded the Nobel Prize in 1996.

As stated by Smalley, "the name [fullerene] was born in the dimmest early thinking of how a pure carbon cluster of 60 atoms could eliminate its dangling bonds" (Billups and Ciufolini, 1993, foreword vi). In an effort to make clear the shape of the cluster, Smalley asked Kroto the name of the architect who worked with big domes. The answer was Buckminister Fuller. Carbon clusters of all sizes were subsequently named Buckminsterfullerenes, fullerenes, or sometimes "buckyballs." A third allotrope of carbon had thus been added to the two (graphite and diamond) already known (see Figure 1).
Fullerenes were available initially only in vanishingly small quantities in the gas phase . An important breakthrough came in 1990 when Wolfgang Kratschmer of the Max Planck Institute for Nuclear Physics and Donald Huffman of the University of Arizona found that fullerenes could be synthesized in gram quantities by electric arc discharge between graphite electrodes immersed in a noble gas . The fullerenes C 60 and C 70 can be isolated readily from the crude soot along with several larger fullerenes. With an abundant supply of these fascinating new materials at hand, a flurry of activity directed at establishing a preliminary picture of fullerene chemistry ensued. The literature is now replete with descriptions of fullerenes that have been isolated using many of the reagents that are available to the organic chemist.
Fullerenes with metals trapped inside their cage may be formed when a graphite target is doped with the metal so that the fullerene grows around the metal. Chemists at Yale University have found that helium can be introduced by heating the fullerene under a high pressure of helium. In this way, a window is formed in the fullerene, which closes as the mixture is cooled, trapping the helium inside the fullerene. The endohedral fullerenes containing metals are promising candidates as magnetic resonance imaging agents.
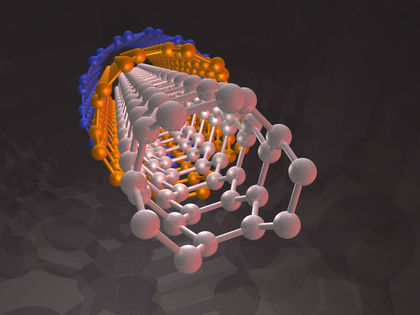
Carbon nanotubes are fullerene structures played out as long strands rather than spheres. In 1993 Sumio Ajima working at the NEC Corporation in Japan discovered that carbon nanotubes could be created using a process similar to the one used by Kratschmer and Huffman to synthesize C 60 . These nanometer-scale structures became the focus of enormous interest since they represent potential building blocks for nanostructured materials, composites, and novel electronic devices of greatly reduced size.
Single-wall carbon nanotubes (SWNTs) can be prepared by laservaporization of a graphite source. A newer process uses carbon monoxide as the source of the carbon and is called the HiPco process. The catalyst is generated in situ from iron carbonyl. The SWNTs from the HiPco process are characterized by a smaller diameter and exhibit greater reactivity with organic reagents.
Since nanotubes are basically rolled-up sheets of graphite, many different tubes with different diameters and structures can be formed. Even a minor difference in the structure of the nanotube can make the material act like a metal or a semiconductor. Semiconducting nanotubes fluoresce upon exposure to light by emitting the light at a different wavelength. Since nanotubes fluoresce differently depending on their structure, it is possible to find an optical signal for each type of tube. In this way, a team of scientists at Rice University have identified thirty-three semiconducting varieties that are formed in the HiPco process, emphasizing the difficulty that researchers face as they attempt to carry out research with these materials. Nevertheless, carbon nanotubes hold great promise as precursors for strong fibers, electrical conductivity of copper and thermal conductivity of diamond, and perhaps even a means of perfecting deoxyribonucleic acid (DNA).
SEE ALSO Allotropes ; Carbon ; Nanochemistry .
W. E. Billups
Bibliography
Ajima, S. (1991). Nature 354:56–58.
Billups, W. E., and Ciufolini, M. A., eds. (1993). Buckminsterfullerenes. New York: VCH.
Kratschmer, W.; Lamb, L. D.; Fostiropoulos, F.; and Huffman, D. (1990). Nature 347:354–358.
Kroto, H. W.; Heath, J. R.; O'Brien, S. C.; Curl, R. F.; and Smalley, R. E. (1985). Nature 318:162–163.
Yakobson, B. I., and Smalley, R. E. (1997). American Scientist 85:324–337.
It gained me a lot of info.