Cosmetic Chemistry

The global market for skincare and color cosmetics exceeded 53 billion dollars in 2002. The number of new products brought to market continues to expand exponentially. Cosmetic chemists are always looking for interesting and exotic ingredients that improve skin's appearance and health. A vast array of compounds is required to supply these products. The latest edition of the Cosmetics Toiletries and Fragrance Association (CTFA) Dictionary lists more than 10,000 raw materials. Every year hundreds of new ingredients are added to the list of those that have been used for centuries. Some materials used today can be traced to 11,000 B.C.E. in the animal drawings from the caves of Altimira.
History
The appearance of skin care formulation dates to around 3000 B.C.E. in ancient Egypt. Most concoctions were prepared from natural materials. Cleopatra is said to have bathed in donkeys' milk to keep her skin smooth and supple. One naturally occurring material used by the ancients was red ochre, or iron oxide. Lumps of red ore were formed when iron oxidized or rusted. The red iron oxide was found in burial tombs in ceremonial lip tints and rouge preparations. It was also used to draw the ancient cave pictures of animals, as seen in Altimira, and is still used in many makeup formulations
today. Eye paints have also been found at ancient gravesites. These paints consisted primarily of a copper-based green ore called malachite that was mined from nearby quarries. Animal fat was combined with fragrant substances such as frankincense and myrrh to produce early skin ointments. More sophisticated creams and lotions were fine tuned through trial and error and passed down over many generations.
Emulsions
The majority of creams and lotions are emulsions. An emulsion can be defined simply as two immiscible fluids in which one liquid is dispersed as fine droplets in the other. Homogenized milk is an example of a typical oil-in-water (o/w) emulsion. Milk fat (oil) is dispersed in water as fine droplets by the homogenization process. The reason the fat does not float to the top immediately is due to the presence of emulsifiers; in this case, a milk protein called sodium caseinate as well as several phopholipids. In the case of water-in-oil (w/o) emulsions, water is dispersed as droplets and suspended in the oil phase. The nondispersed liquid or external suspending phase is also called the continuous phase. Mayonnaise, vinegar water dispersed as fine droplets in a continuous phase of soybean oil, is an example of a water-in-oil emulsion. Lecithin from eggs stabilizes the mayonnaise emulsion.
Surfactants
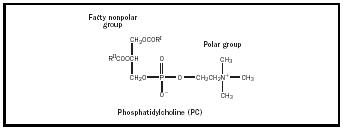
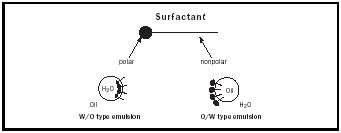
Most emulsifiers can be considered surfactants or surface-active agents. These materials are able to reduce the surface tension of water. What makes an emulsifier surface active is related to its HLB, or hydrophile-lipophile balance. HLB is determined by the size of the hydrophilic (water-loving or polar) portion of a molecule as compared to the size of the lipophilic (oilloving or nonpolar) portion. The HLB system was created to rank the relative polarity of materials. The most polar, water soluble, materials are at the top of the twenty-point scale with more non-polar, oil soluble, materials closer to zero. The HLB of sodium caseinate is assigned a value of around fourteen because of it's high solubility in water. Lecithin, being poorly soluble in water, has an HLB value of about six. Both have polar groups. The polar group in the milk protein is sodium. Lecithin's surface-active component is a molecule called phosphotidylcholine or PC (See Figure 1). The polar, or water soluble part of PC is the phosphate functional group. The emulsifiers' polar groups orient toward the polar water phase. Their lipophilic, nonpolar groups oriented toward the oil phase to form micelles (see Figure 2). These spherical structures provide stability to the emulsion through Hydrogen bonding and weak electrical forces.
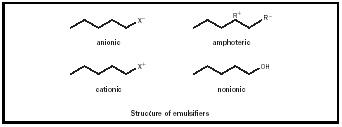
Skin-care emulsifiers can be divided into two groups based on ionic charge (See Figure 3). Materials that can dissociate into charged species are considered ionic while those that do not are called nonionic. Ionic emulsifiers can be further classified by type of charge. Anionics are negatively charged when solvated as in sodium stearate or soap.
When fatty acids are reacted with alkali they form soaps. The process of soap formation is called saponification. The negatively charged stearic acid group is the main emulsifying unit of the soap, giving it the anionic classification. Positively charged emulsifiers are called cationic. Quarternium24's emulsifying unit dissociates into the positively charged ammonium group. Amphoterics are compounds that express both negative and positive charges.
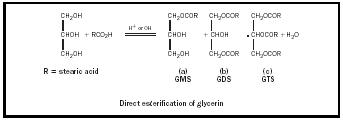
Nonionic emulsifiers are often used in skin-care emulsion for their safety and low reactivity. They are generally classified by chemical similarity. Glycerin, commonly added to cosmetic emulsions for its humectant properties, is the backbone of a class of emulsifiers called Glyceryl esters . Glyceryl monostearate, or GMS, is called a monoester because of its sole ester linkage (see Figure 4). The diester is prepared by esterifying two molecules of stearic acid for every molecule of glycerin. Glyceryl mono- and diesters are very effective emulsifiers because they contain both polar hydroxyl (OH) groups as well as non-polar fatty acids. If all three of Glycerin's hydroxyl groups are reacted, the resulting triester will have little emulsifying capability.
Stearic acid is called C18 fatty acid. The fatty acids, present in fats and oils, are classified according to their carbon-chain lengths. Because stearic acid is a major component of many of the fats and oils used in beauty treatments, stearate-based emulsifiers are particularly useful. Fatty acids are key components of many cosmetic emulsifiers due their miscibility in a variety of natural and synthetic oils.
Esters polyethylene glycol or ethylene glycol are called PEG esters. A PEG ester's solubility is determined by the number of PEG molecules reacted per molecule of acid. PEG 6 oleate for instance has six molecules of PEG reacted with one molecule of oleic acid. As the number of polar, PEG molecules per acid molecule increases the water solubility/HLB is increased; PEG 8 oleate is more soluble than PEG 6 oleate. The cosmetic chemist will often use blends of glyceryl esters and a PEG ester with high and low HLB values to determine the required polarity to emulsify various fats and oils. The many types of emulsifiers are too numerous to list here, however McCutcheon's Emulsifiers and Detergents is an excellent source for a more complete listing.
Emollients
The majority of emollients used in personal care and beauty items are fats and oils, also called lipids. Animal fat or tallow is composed primarily of stearic and palmitic acids with carbon chains lengths of 18 and 16 respectively. Many of the major cosmetic companies are moving away from animal-based materials like tallow to renewable vegetable-based materials. Coconut oil and palm kernel oil are often used. Some of the key characteristics required in good emollients are good spreading properties, low toxicity/skin irritation and good oxidative stability. Oleic acid, a major constituent of olive oil has poor oxidative stability due to the presence of its double bond. Fats and oils are considered saturated if they do not have double bonds. Unsaturated oils like olive oil have double bonds that can react with oxygen, especially when heated. The oxidation process can produce off colors and odors in lipids causing them to go rancid and unusable.
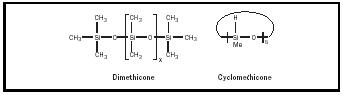
Petroleum-based emollients such as petroleum jelly and mineral oil are found in many formulations because they do not contain double bonds or reactive functional groups. Silicone oils such as cyclomethicone, dimethicone are often added to increase slip and emolliency (See Figure 5).
Oils that contain high levels of essential fatty acids, EFAs, are prized for their ability to replenish lipids (oils) that are found naturally within the skin layers. Linoleic acid is an example of an EFA. Long-chain alcohols, also called fatty alcohols, are useful as emollients and emulsion stabilizers. Their polar hydroxyl groups orient to the water phase with their fatty chains oriented towards the oil phase. Esters of fatty alcohols and fatty acids make excellent emollients because of their low reactivity and good stability.
Lanolin, derived from sheep's wool, is often called wool grease. Lanolin has been used for centuries due to it's unique composition of complex sterols, fatty alcohols, and fatty acids. Cholesterol, a cyclic molecule called
a sterol, is a major component. The polar hydroxyl groups of sterols and alcohols enable the grease to absorb and hold water. Skin is primarily composed of water, countless oils and emollients are used to nourish and protect it.
Moisturizers
The main distinction between moisturizers and emollients is their solubility in water. Healthy skin requires moisture. Moisturizers are generally polar materials that are hygroscopic in nature; they hold onto water. An important tool to assess the efficiency of moisturizers is the high scope. It measures transepidermal water loss or TEWL. After a moisturizer is applied to the skin, the moisture level is recorded. After several minutes the moisture level will be reduced due to the natural tendency of the skin to release moisture over time. Ingredients that can maintain a high level of moisture in the upper layers of the skin for several hours can reduce the rate at which water is lost. Glycerin is a very cost-effective ingredient used to help reduce TEWL. Sorbitol, sucrose, glucose, and other sugars are also commonly used to hydrate the skin. Aloe, which contains a mixture of polysaccharides, carbohydrates, and minerals, is an excellent moisturizer. As skin becomes drier in the winter months, it may be necessary to incorporate materials that better seal the moisture in the skin.
Waxes
Waxes are composed primarily of long-chain esters that are solid at room temperature. Anyone who has ever dipped a finger in molten wax has experienced its sealing properties. Some common waxes used in cosmetics are beeswax, candelilla, carnauba, polyethylene, and paraffin. The melting points of waxes vary widely depending on their unique composition and chain lengths. Commonly used in lip balms and sticks, waxes function as structuring agents, giving the stick enough rigidity to stand up on its own, as well as barrier properties. By combining waxes with different properties such as high shine, flexibility, and brittleness, optimal cosmetic performance can be achieved. Often waxes are combined with compatible oils to achieve the desired softness. Compatibility is generally determined by gauging the turbidity and degree of separation of two materials mixed together above their melt points. Waxes are particularly useful in hand creams and mascara emulsions for their thickening and waterproofing properties.
Thickeners
By incorporating enough wax into a thin lotion, a thick cream can be formed. Many thickeners are polymers. Cellulose, a fine powder polymer of repeating
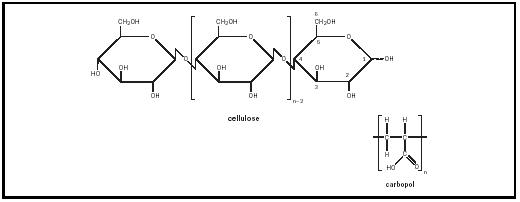
D-glucose units, swells in hot water creating a gel network. Carbopol, a polyacrylic acid, swells when neutralized (See Figure 6). Bentone clays swell when their structure, resembling a stack of cards, is opened up through mechanical shear. Carrageenan, pectin, and locust bean gum are all examples of cosmetic thickeners that are also used in some of our favorite foods such as jellies, salad dressings, and pie fillings.
Active Ingredients
Materials that work physiologically within the skin or aid in protecting the skin from insult are also called active ingredients. The term "cosmeceuticals" coined by famed dermatologist Dr. Albert Kligman, refers to a product that is in-between a cosmetic and a drug. Although a cosmetic, by legal definition, can only serve to beautify and protect the surface of the skin, many cosmetic products can be shown to penetrate the dermal layers of the skin to exact a physiological change.
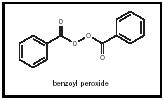
Fruit acids are an example of an active material. Also called alpha hydroxyacids or AHAs, they have the ability to penetrate the skin, where they can increase the production of collagen, elastin, and intracellular substances thus improving the appearance of the skin. Thousands of cosmetic actives are used to affect the skin in a variety of ways. They are used to lighten, tighten, and firm the skin. They can be used to suppress perspiration as in the case of aluminum chlorohydrate. Salicylic acid and benzoyl peroxide are important ingredients because of their anti-acne activity (See Figure 7). Some active materials are added to skin treatments to protect the skin from the environment. Dimethicone and petrolatum are examples of skin protectants.
Sunscreens
Sunscreens are a class of compounds that protect the skin from ultraviolet radiation. Wavelengths between 290nm and 400nm are particularly damaging to the skin. Sunscreens' ability to absorb or reflect these damaging wavelengths are rated by their SPF or sun-protection factor. For instance a person protected with a factor-15 sunscreen will be able to stay in the sun fifteen times longer than if unprotected. Octyl methoxycinnamate, octyl salycilate, titanium dioxide, and avobenzone are some important topical sunscreens. They can be classified as either UVA or UVB sunscreens depending the wavelengths they absorb. Benzophenone 4, a water-soluble UV filter, is commonly used to protect the color of cosmetic products.
Color
Pigments and dyes are used in products to impart a color. Titanium dioxide (TiO 2 ) is a white pigment that is mined. In combination with natural mined and synthetic iron oxides, which range in color from red, yellow, black and brown, depending upon the degree of oxidation and hydration, a range of color can be produced that will be suitable for almost every skin tone. Face powders are produced by blending inorganic oxides and fillers. Fillers are inert, generally inexpensive materials such as kaolin, talc, silica, and mica that are used to extend and fully develop colors. Pressed powders like eyeshadows and blushers are prepared by blending additional binding ingredients such as oils and zinc stearate and pressing the mixture into pans.
Eyeshadows and lipsticks often contain pearlescent pigments commonly called pearls. Pearls sparkle and reflect light to produce a multitude of colors. They are prepared by precipitating a thin layer of color on thin platelets of mica. Varying the thickness of the color deposited changes the angle of light refracted though the composite, creating different colors.
Organic pigments are used to color lipsticks and eyeshadows. When organics are precipitated on a substrate they are called lake pigments. The term lake refers to the laking or precipitating of the organic salt onto a metal substrate such as aluminum, calcium, or barium. They are called D&C (drug and cosmetic) and FD&C (food, drug and cosmetic) colors. Some examples are D&C Red#7 calcium lake and FD&C Yellow #5 aluminum lake. Dyes such as FD&C Blue#1 and D&C Yellow #10 are readily soluble as opposed to pigments, which are insoluble. Dyes are useful in providing tints for lotions, oils, and shampoos.
Preservatives
Most cosmetic products require the addition of preservative to prevent microbial contamination and rancidity. Parabens and ester of parabenzoic acid are by far the most commonly used because of their effectiveness against gram-positive bacteria. Phenoxyethanol is used to protect against gramnegative strains. The cosmetic chemist will generally employ a mixture of preservatives to protect against different bacterial strains as well as yeasts and molds. Antioxidants such as tocopherol (vitamin E) and BHT are also added to prevent oxidation of sensitive ingredients as well as protect the skin from free-radical damage.
Conclusion
As long as society continues to puts great emphasis on looking young and beautiful cosmetic chemistry will continue to flourish. A good understanding of the fundamentals of emulsion chemistry and skin physiology are prerequisites, when formulating personal care products. A good cosmetic chemist must be able to combine science and art to create products with the feel and look that consumers desire.
John Castro
Bibliography
Harry, Ralph G. (2000). Harry's Cosmeticology , 8th edition, ed. Martin M. Reiger. Chemical Publishing Company.
International Cosmetic Ingredient Dictionary and Handbook , 9th edition. (2002). Washington, DC: Cosmetic, Toiletry, and Fragrance Association.
Lewis, Peter A. (1988). Pigment Handbook, Volume 1 , 2nd edition. Hoboken, NJ: Wiley Interscience.
Wan, Peter J. (1991). Introduction to Fats and Oils Technology. Champaign, IL: The American Oil Chemists' Society.
Thank you.
(But I guess you never did a Mayonnaise, isn't it? You would know it's an o/w emulsion.)