Inorganic Industrial Chemistry

Industrial inorganic chemistry includes subdivisions of the chemical industry that manufacture inorganic products on a large scale such as the heavy inorganics (chlor-alkalis, sulfuric acid, sulfates) and fertilizers (potassium, nitrogen, and phosphorus products) as well as segments of fine chemicals that are used to produce high purity inorganics on a much smaller scale. Among these are reagents and raw materials used in high-tech industries, pharmaceuticals or electronics, for example, as well as in the preparation of inorganic specialties such as catalysts, pigments, and propellants.
Metals are chemicals in a certain sense. They are manufactured from ores and purified by many of the same processes as those used in the manufacture of inorganics. However, if they are commercialized as alloys or in their pure form such as iron, lead, copper, or tungsten, they are considered products of the metallurgical not chemical industry.
The Chemical Industry
The chemical industry adds value to raw materials by transforming them into the chemicals required for the manufacture of consumer products. Since there are usually several different processes that can be used for this purpose, the chemical industry is associated with intense competition for new markets. It is made up of companies of different sizes, including several giants that are engaged in the transformation of some very basic raw materials into final products, as well as medium-size or small companies that concentrate on very few of these steps. The closer to the raw material, the larger the scale of operations; such "heavy" inorganic chemicals are usually manufactured by continuous processes. At the other extreme in terms of scale are the firms that manufacture "specialties," mostly in batch processes, from "intermediates" that correspond to chemicals which have already gone through several steps of synthesis and purification.
Basic chemicals represent the starting point for the manufacture of inorganic industrial chemicals. They are usually one step away from the raw materials listed in Table 1 and are produced on a very large scale employing continuous processes. The unit price of these products is relatively low, and producing them cheaply and efficiently is a major concern for the companies that manufacture them. Sulfur, nitrogen, phosphorus, and chloralkali industries are the main producers of basic inorganic chemicals, and they will often sell them to other industries as well as using them in the manufacture of their own end-products. The basic principles for their production and major uses are indicated here for each of these industries.
Inorganic chemicals produced on an industrial scale can be easily identified. Many of today's large companies started as producers of inorganics,

but as coal—and especially petroleum—became important sources of raw materials, they were integrated into the product chain.
Sources of Inorganic Raw Materials
There are many different sources of raw materials for the manufacture of inorganic chemicals (see Table 1). Very few of them are found in their elemental form. Sulfur is a notable exception. It occurs in underground deposits and can be brought to the surface by compressed air after it is melted by superheated steam. However, increasing quantities of sulfur are recovered from petroleum and natural gas (where they occur as impurities).
Air contains molecular nitrogen and oxygen. They may be separated by liquefaction and fractional distillation along with inert gases, especially argon. Salt or brine can be used as sources of chlorine and sometimes bromine, sodium hydroxide, and sodium carbonate, whereas metals such as iron, aluminum, copper, or titanium as well as phosphors, potassium, calcium, and fluorine are obtained from mineral ores. Saltpeter was once an important source of nitrogen compounds, but today most ammonia and nitrates are produced synthetically from nitrogen gas in the air.
Recovery and recycling provide increasing amounts of some metals. As environmental concerns increase, these operations will probably become an important source of materials used in the manufacture of certain inorganic chemicals.
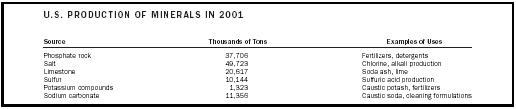
| U.S. PRODUCTION OF MINERALS IN 2001 | ||
| Source | Thousands of Tons | Examples of Uses |
| Phosphate rock | 37,706 | Fertilizers, detergents |
| Salt | 49,723 | Chlorine, alkali production |
| Limestone | 20,617 | Soda ash, lime |
| Sulfur | 10,144 | Sulfuric acid production |
| Potassium compounds | 1,323 | Caustic potash, fertilizers |
| Sodium carbonate | 11,356 | Caustic soda, cleaning formulations |
The Beginnings of the Chemical Industry
The origins of the chemical industry can be traced to the Industrial Revolution . Sulfuric acid and sodium carbonate were among the first industrial chemicals. "Oil of vitriol " (as the former was known) played an important role in the manipulation of metals, but its production on an industrial scale required the development of materials that would resist attack. Sodium carbonate was obtained in its anhydrous form, "soda ash," from vegetable material until the quantities produced could no longer meet the rapidly expanding needs of manufacturers of glass, soap, and textiles. This led the Royal Academy of Sciences of Paris, in 1775, to establish a contest for the discovery of a process based on an abundant raw material, sodium chloride, and to Nicolas Leblanc's method for the preparation of soda by converting salt into sulfate
2 NaCl + H 2 SO 4 → Na 2 SO 4 + 2 HCl
followed by conversion of the sulfate to soda with charcoal and chalk
Na 2 SO 4 + 2 C + CaCO 3 → Na 2 CO 3 + CaS + 2 CO 2
Although he did not win the prize, Leblanc's process is associated with the birth of industrial chemistry.
The industrial production of chemicals was usually based on running reactions that were known to yield the desired products on much larger scales. Success in these endeavors lay much more in the experience and skill of their practitioners than the application of solid chemical principles. This led to serious problems of control and the generation of noxious by-products. The introduction of the Leblanc process in the northwest of England led to a general public outcry against the dark and corrosive smoke that covered the surrounding countryside. The Alkali Act, passed in response in 1863, represents the first legislation that established emission standards.
Sulfuric acid was an essential chemical for dyers, bleachers, and alkali manufacturers. Its production on a large scale required the development of lead-lined chambers that could resist the vapors which were formed when sulfur was burned with nitrates
SO 2 + NO 2 + H 2 O → H 2 SO 4 + NO
NO + 1/2 O 2 → NO 2
This process was wasteful and emitted corrosive gases . It improved only in the mid-nineteenth century when towers to recycle the gases were finally introduced. The transportation of sulfuric acid was dangerous, and alkali manufacturers tended to produce their own as a result. This marked the beginning of the diversification and vertical integration that are characteristic of the chemical industry.
Sulfuric acid was also used in the manufacture of superphosphates, which were produced as fertilizers on a large scale by the mid-nineteenth century. By that time, a solution was found for the complex engineering problems that had hampered the use of the alternative process to produce soda
NH 3 + H 2 O + CO 2 → NH 4 HCO 3
NaCl + NH 4 HCO 3 → NaHCO 3 + NH 4 Cl
2 NaHCO 3 → Na 2 CO 3 + H 2 O + CO 2
Ernest Solvay, a Belgian chemist, designed a tower in which carbon dioxide reacted efficiently with solid salts. The Solvay process had enormous advantages over the Leblanc process: It did not generate as much waste and pollution; its raw materials, brine and ammonia, were readily available (the latter from gasworks); less fuel was used, and no sulfur or nitrate was involved. In spite of its higher capital costs, it was rapidly adopted and soon became the major source of alkali.
Another major process used in the manufacture of inorganic chemicals is the catalytic conversion of nitrogen and hydrogen to ammonia. The German chemist Fritz Haber first synthesized ammonia from nitrogen and hydrogen in 1909. Four years later, together with another German, Carl Bosch, he modified the process for the commercial production of ammonia. The Haber (or Haber–Bosch) process represented a technological breakthrough since it required a very specialized plant to handle gases at high pressures and temperatures.
Sulfuric Acid and Sulfates
Sulfuric acid has long been the chemical that is manufactured in the largest quantities on a world scale (see Table 2). Its production is often linked to a country's stage of development, owing to the large number of transformation processes in which it is used.
Sulfuric acid is manufactured from elemental sulfur. Mining was the main source for this element, which was obtained from sulfide-containing ores, or in very pure form from underground deposits by the Frasch process (injection of superheated steam and air into drillings and the separation of the mixture that rises to the surface). The large-scale consumption of petroleum and natural gas has changed this scenario since sulfur occurs as an impurity in most fossil fuels and must be removed before the fuels are processed. These fuels are presently the main source of sulfur, and their relative importance tends to increase with more rigorous controls on emissions.
Sulfuric acid is manufactured in three stages
2 SO 2 + O 2 → 2SO 3
SO 3 + H 2 O → H 2 SO 4
Since the reaction of sulfur with dry air is exothermic, the sulfur dioxide must be cooled to remove excess heat and avoid reversal of the reaction.
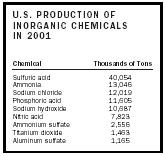
| U.S. PRODUCTION OF INORGANIC CHEMICALS IN 2001 | |
| Chemical | Thousands of Tons |
| Sulfuric acid | 40,054 |
| Ammonia | 13,046 |
| Sodium chloride | 12,019 |
| Phosphoric acid | 11,605 |
| Sodium hydroxide | 10,687 |
| Nitric acid | 7,823 |
| Ammonium sulfate | 2,556 |
| Titanium dioxide | 1,463 |
| Aluminum sulfate | 1,165 |
Most plants use reactors with various stages in order to cool the stream for the catalytic step. Conversion by a vanadium pentoxide catalyst deposited on a silicate support is the critical step in the process, in which the gaseous stream is passed over successive layers of catalyst. The gas mixture is then passed through an absorption tower. Oleum, the product, is a concentrated solution of sulfuric acid containing excess sulfur trioxide.
As an inexpensive source of acid, a large amount of the sulfuric acid that is produced is used for the manufacture of other mineral acids. It is also used to produce sulfates, such as ammonium sulfate (a low-grade fertilizer), sodium sulfate (used in the production of paper), and aluminum sulfate (used in water treatment), as well as organic sulfates (used as surfactants ). Sulfuric acid is also a good catalyst for many reactions, including the transformation of ethanol into ethylene or ethyl ether.
Nitrogen Compounds
In general, chemicals containing nitrogen are manufactured from ammonia produced by the Haber process
Since molecular nitrogen is inert, its reaction with hydrogen requires very severe conditions and a catalyst. An iron catalyst is used. High pressure favors the formation of products, but an increase in temperature will shift the equilibrium in the opposite direction. Plants will thus operate under conditions that represent the most favorable balance between operating costs and capital investment.
Energy consumption is very high, and its cost is an important component along with the starting materials. Nitrogen is easily obtained from air, and hydrogen and can be produced by the shift reaction
CO + H 2 O → CO 2 + H 2
or from hydrocarbon reforming
CH 4 + 2H 2 O → CO 2 + 4H 2
Further stages are required to assure conversion and to remove carbon dioxide or carbon monoxide from the gas mixture. A mixture of ammonia and synthesis gas (CO + H 2 ) results from the reaction with nitrogen so the two must be separated and the synthesis gas recycled.
Most of the ammonia that is produced is employed as fertilizer or used to manufacture other fertilizers, such as urea, ammonium sulfate, ammonium nitrate, or diammonium hydrogen phosphate. Ammonia is also used in the Solvay process, and it is a starting material for the manufacture of cyanides and nitriles (which are used to make polymers such as nylon and acrylics) as well as aromatic compounds containing nitrogen, such as pyridine and aniline.
The other source of nitrogen compounds in the chemical industry is nitric acid, obtained from the oxidation of ammonia
4 NH 3 + 5 O 2 → 4 NO + 6 H 2 O
3 NO + 3/2 O 2 → 3 NO 2
3 NO 2 + H 2 O → 2 HNO 3 + NO
The first reaction is run over platinum-rhodium catalysts at around 900°C (1,652°F). In the second and third stages, a mixture of nitric oxide and air circulates through condensers, where it is partially oxidized. The nitrogen dioxide is absorbed in a tower, and nitric acid sinks to the bottom. Nitric acid is mainly used to make ammonium nitrate, most of it for fertilizer although it also goes into the production of explosives. Nitration is used to manufacture explosives such as nitroglycerine and trinitrotoluene (TNT) as well as many important chemical intermediates used in the pharmaceutical and dyestuff industries.
Phosphorus Compounds
The world's major source of phosphorus is apatite, a class of phosphate minerals. Commercially, the most important is fluoroapatite, a calcium phosphate that contains fluorine. This fluorine must be removed for the manufacture of phosphoric acid, but it also can be used to produce hydrofluoric acid and fluorinated compounds.
Phosphoric acid is the starting material for most of the phosphates that are produced industrially. It is obtained from the reaction of the apatite mineral with sulfuric acid
Silica is present in the mineral as an impurity, and it reacts with hydrofluoric acid to yield silicon tetrafluoride, which can be converted to fluorosilicic acid, an important source of fluorine. More than half of the phosphoric acid that is produced by the reaction of phosphates with sulfuric acid is converted directly to sodium or ammonium phosphates to be used as fertilizer; thus, purity is not a concern.
For products that require high purity, such as detergents and foodstuffs, phosphoric acid is produced from elemental phosphorus (at about four times the cost). An electric furnace operating at 1,400–1,500°C (2,552–2,732°F) is used to form a molten mass of apatite and silica that reacts with coke and reduces the phosphate mineral
2 Ca 3 (PO 4 ) 2 + 6 SiO 2 + 10 C → P 4 + 6 CaSiO 3 + 10 CO
Concentrating phosphoric acid leads to polyphosphoric acid, a mixture of several polymeric species, a good catalyst and dehydrating agent. Polyphosphate salts are used as water softeners in detergents or as buffers in food. Small quantities of elemental phosphorus are used to make matches, and phosphorus halides to prepare specialty chemicals for the pharmaceutical and agrochemical industries.
Chlor-Alkali Compounds
Industries producing chlorine, sodium hydroxide (also known as caustic soda), sodium carbonate (or soda ash) and its derivatives and compounds based on calcium oxide (or lime) are usually included under this category. As both sodium hydroxide and chlorine have a common raw material, sodium chloride, they are produced in quantities that reflect their equal molar ratio, irrespective of the market for either product. Since they are produced by electrolysis, they require a cheap source of brine and electricity
2 NaCl + 2 H 2 O → 2 NaOH + Cl 2 + H 2
Most processes are based on the electrolysis of a sodium chloride solution, but some plants operate with the molten salt. Three different cell types are used in electrolysis in water: mercury cells, diaphragm cells, and membrane cells. Membrane cells are replacing the other two types in modern units, but it may not be economically feasible to convert older plants.
Sodium hydroxide and sodium carbonate are alternative sources of alkali, and their use has followed the availability of raw materials as well as the efficiency of processes developed for their production. Both require sodium chloride and energy and, if limestone deposits are also available, sodium carbonate may be produced by the Solvay process. Limestone consists mainly of calcium carbonate and can be used to produce calcium oxide (or quicklime) and calcium hydroxide (or slaked lime); the oxide may be obtained by heating (1,200–1,500°C, or 2,192–2,732°F) limestone, while the hydroxide, which is more convenient to handle, is obtained by adding water to the oxide
CaO + H 2 O → Ca(OH) 2
Its principal use is in steelmaking, but it also goes into the manufacture of chemicals, water treatment, and pollution control. In the Solvay process, calcium carbonate and sodium chloride are used to produce calcium chloride and sodium carbonate with ammonia (which is recycled) as a medium for dissolving and carbonating the sodium chloride and calcium hydroxide for precipitating calcium chloride from the solution.
As sodium carbonate may be mined directly, its use may be preferred over a manufactured product. It is used mainly in the glass industry. Sodium silicates may be derived from sodium carbonate and in their finely divided form, silica gel, may be used in detergents and soaps.
Sodium hydroxide has many different uses in the chemical industry. Considerable amounts are used in the manufacture of paper and to make sodium hypochlorite for use in disinfectants and bleaches. Chlorine is also used to produce vinyl chloride, the starting material for the manufacture of polyvinyl chloride (PVC), and in water purification. Hydrochloric acid may be prepared by the direct reaction of chlorine and hydrogen gas or by the reaction of sodium chloride and sulfuric acid. It is used as a chlorinating agent for metals and organic compounds.
In certain regions of the world, there are salt deposits or brines that have been enriched by bromine. Commercially, bromine may be extracted by treating the brines with chlorine and removing it by steam.
2 Br − + Cl 2 → Br 2 + 2Cl −
Bromine is used in water disinfection; bleaching fibers and silk; and in the manufacture of medicinal bromine compounds and dyestuffs.
Titanium Dioxide
Titanium dioxide is by far the most important titanium compound. It can be purified by dissolving in sulfuric acid and precipitating the impurities. The solution is then hydrolyzed, washed, and calcinated . Alternatively, ground rutile is chlorinated in the presence of carbon and the resulting titanium tetrachloride is burned in oxygen to produce the chloride.
Titanium dioxide is found in nature in three crystal forms: anastase, brookite, and rutile. Its extreme whiteness and brightness and its high index of refraction are responsible for its widespread use as a white pigment in paints, lacquers, paper, floor covering, plastics, rubbers, textiles, ceramics, and cosmetics.
SEE ALSO Aluminum ; Catalysis and Catalysts ; Copper ; Fertilizer ; Haber, Fritz ; Iron ; Steel .
Peter Rudolf Seidl
Carlos Eduardo da Silva Cortes
Bibliography
Brock, William H. (2000). The Chemical Tree: A History of Chemistry. New York: Norton.
Budavari, Susan, ed. (1996). The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 12th edition. Whitehouse Station, NJ: Merck.
Heaton, Alan, ed. (1994). The Chemical Industry, 2nd edition. New York: Blackie Academic & Professional.
Heaton, Alan, ed. (1996). An Introduction to Industrial Chemistry, 3rd edition. New York: Blackie Academic & Professional.
Comment about this article, ask questions, or add new information about this topic: