Glass
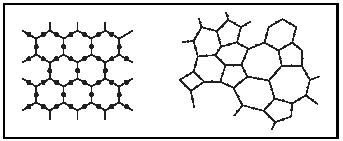
Glass is a state of matter. It is a solid produced by cooling molten material so that the internal arrangement of atoms, or molecules, remains in a random or disordered state, similar to the arrangement in a liquid. Such a solid is said to be amorphous or glassy. Ordinary solids, by contrast, have regular crystalline structures. The difference is illustrated in Figure 1.
Many materials can be made to exist as glasses. Hard candies, for example, consist primarily of sugar in the glassy state. What the term "glass" means to most people, however, is a product made from silica (SiO 2 ). The
common form of silica is sand, but it also occurs in nature in a crystalline form known as quartz.
Pure silica can produce an excellent glass, but it is very high-melting (1,723 o C, or 3,133 o F), and the melt is so extremely viscous that it is difficult to handle. All common glasses contain other ingredients that make the silica easier to melt and the hot liquid easier to shape.
Natural Glass
Probably as early as 75,000 B.C.E. , long before human beings had learned how to make glass, they had used natural glass to fashion knives, arrowheads, and other useful articles. The most common natural glass is obsidian, formed when the heat of volcanoes melts rocks such as granite, which then become glassy upon cooling. Other natural glasses are pumice, a glassy foam produced from lava; fulgurites, glass tubes formed by lightning striking sand or sandy soil; and tektites, lumps or beads of glass probably formed during meteoric impacts.
Manmade (Synthetic) Glass
When, where, or how human beings discovered how to make glass is not known. Very small dark-colored beads of glass have been dated back to 4000 B.C.E. These may well have been by-products of copper smelting or pottery glazing. By 2500 B.C.E. small pieces of true synthetic glass appeared in areas such as Mesopotamia, but an actual glass industry did not appear until about 1500 B.C.E. in Egypt. By this time various small vases, cosmetic jars, and jewelry items made of glass had begun to appear.
All the ancient glasses were based on silica (sand), modified with considerable amounts of various metal oxides, mainly soda (Na 2 O) and lime (CaO). This is still the most common glass being used today. It is known as soda lime glass. However, the ancient glass was usually colored and opaque due to the presence of various impurities, whereas most modern glass has the useful property of transparency.
Hundreds of thousands of different glass compositions have been devised, and they have been used in different ways. Much has been learned about which combination of chemicals will make the best glass for a particular purpose. For example, in 1664 an Englishman named Ravenscroft found that adding lead oxide (PbO) to a glass melt produced a brilliant glass that was much easier to melt and to shape. Since that time lead glass has been used to make fine crystal bowls and goblets and many kinds of art glass.
An important kind of glass was developed in the early 1900s to solve a serious problem—the inability of glass to withstand temperature shock. This failure resulted in tragic accidents in the early days of the railroads. Glass lanterns used as signals would get very hot, and then, if it started to rain, the rapid cooling would sometimes cause the glass to break and the signal to fail. The problem was solved by replacing much of the soda in the glass with boron oxide (B 2 O 3 ). The resulting glass, called borosilicate, contains about 12 percent boron oxide and can withstand a temperature variation of 200 o C (392 o F). It also has greater chemical durability than soda lime glass. Today borosilicate glass is used in most laboratory glassware (beakers, flasks, test tubes, etc.) and in glass kitchen bakeware.
For even greater heat shock resistance and chemical durability, alumina (Al 2 O 3 ) can be used instead of boron oxide. The resultant aluminosilicate glass has such resistance to heat shock that it can be used directly on the heating element of the kitchen stovetop. It is also used to make the special bottles used for liquid pharmaceutical prescriptions, and to produce the glass thread that is woven into fiberglass fabric.
High silica glass (96.5–100% silica) remains difficult to make because of the very high melting point of pure silica. However, it is made for special purposes because of its outstanding durability, excellent resistance to thermal shock or chemical attack, and ability to transmit ultraviolet light (an ability that ordinary glass does not have). Spacecraft windows, made of 100 percent silica, can withstand temperatures as high as 1,200 o C (2,192 o F). Table 1 lists the five major types of glass along with properties and uses.
Glass Composition. The making of glass involves three basic types of ingredients: formers, fluxes, and stabilizers. The glass former is the key component in the structure of a glassy material. The former used in most glasses is silica (SiO 2 ). Pure silica is difficult to melt because of its extremely high melting point (1,723 o C, or 3,133 o F), but fluxes can be added to lower the melting temperature. Other glass formers with much lower melting points (400 o C–600 o C, or 752–1,112 o F) are boric oxide (B 2 O 3 ) and phosphorus pentoxide (P 2 O 5 ). These are easily melted, but because their glass products dissolve in water, they have limited usefulness.
Most silica glasses contain an added flux, so that the silica can be melted at a much lower temperature (800 o C–900 o C, or 1,472–1,652 o F). Standard fluxes include soda (Na 2 O), potash (K 2 O), and lithia (Li 2 O). Frequently the flux is added as a carbonate substance (e.g., Na 2 CO 3 ), the CO 2 being driven off during heating. Glasses containing only silica and a flux, however, have poor durability and are often water-soluble.
To make glasses stronger and more durable, stabilizers are added. The most common stabilizer is lime (CaO), but others are magnesia (MgO), baria (BaO), and litharge (PbO). The most common glass, made in largest amounts by both ancient and modern glassmakers, is based on silica as the glass former, soda as the flux, and lime as the stabilizer. It is the glass used to make windows, bottles, jars, and lightbulbs.
Colored Glass. The natural glasses used by the ancients were all dark in color, usually ranging from olive green or brown to jet black. The color was
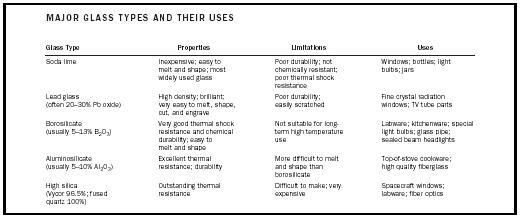
| MAJOR GLASS TYPES AND THEIR USES | |||
| Glass Type | Properties | Limitations | Uses |
| Soda lime | Inexpensive; easy to melt and shape; most widely used glass | Poor durability; not chemically resistant; poor thermal shock resistance | Windows; bottles; light bulbs; jars |
| Lead glass (often 20–30% Pb oxide) | High density; brilliant; very easy to melt, shape, cut, and engrave | Poor durability; easily scratched | Fine crystal radiation windows; TV tube parts |
| Borosilicate (usually 5–13% B 2 O 3 ) | Very good thermal shock resistance and chemical durability; easy to | Not suitable for long-term high temperature use melt and shape | Labware; kitchenware; special light bulbs; glass pipe; sealed beam headlights |
| Aluminosilicate (usually 5–10% Al 2 O 3 ) | Excellent thermal resistance; durability | More difficult to melt and shape than borosilicate | Top-of-stove cookware; high quality fiberglass |
| High silica (Vycor 96.5%; fused quartz 100%) | Outstanding thermal resistance | Difficult to make; very expensive | Spacecraft windows; labware; fiber optics |
due to the presence of significant amounts of metal impurities, especially iron. Even today the ubiquitous presence of iron in nature causes most ordinary glass to have a slight greenish cast.
Many standard glass colorants are oxides of metals such as cobalt (blue), chromium (green), and manganese (violet). Yellow glass is usually made with cadmium sulfide, and red or pink glass usually contains selenium, although some ruby-colored glass has had gold added. The coloring of glass is not a simple subject. Glass color depends not only on which elements are added, but also on the composition of the glass, and on whether the furnace used was in an oxidizing or reducing mode. Copper, for example, can produce blue, green, or opaque red glass, depending on melting conditions.
The Egyptians of 1500 B.C.E. knew that they could make brightly colored glasses by adding certain metals (or their compounds) to the glass melt. The ancient Romans continued the science of making colored glass and expanded it. By the fourth century C.E. the Romans had learned how to produce a dichroic (two-color) glass. The most famous dichroic glass article left by the Romans is the Lycurgus Cup (now at the British Museum). It is green in reflected light (with the lamp in front of the cup), but red in transmitted light (the lamp behind the cup). This unusual glass contains microscopic particles of gold and silver.
Glass Forming
Ancient Methods. Shaping hot, molten glass into useful articles has long been a challenge. Molten glass is extremely hot, caustic, sticky, and difficult to handle. In the period extending from about 2000 B.C.E. to 50 B.C.E. , there were three basic methods used to form glass. One of the earliest and most widely used was core forming. This involved distributing molten glass around a clay core on a metal rod. The rod with the clay core could either be dipped into molten glass, or the hot liquid glass could simply be poured over it. The outer glass coating was then rolled (marvered) on a flat stone surface to smooth it. Often the object was decorated by dribbling more glass, sometimes of a different color, onto its surface. The hot glass was then annealed (cooled slowly so as to relieve thermal stress), and the metal rod was removed and the clay core scraped out.
A second method involved sagging and fusing. It called for taking preformed glass rods or canes (which were often of different colors), placing them in or on top of a mold, and then heating the canes until they sagged and fused together and conformed to the shape of the mold. (Sheets of glass could also be sagged over shaped clay molds.)
The third method was casting, which called for pouring hot, molten glass into a mold. A variation on cast glass was faience, made from powdered quartz blended into molten glass. The mixture might be pressed between two molds to make a cast vessel such as a bowl.
All three of these methods were slow, and they generally produced small items that were rather thick. Glass pieces tended to be quite expensive, and, in antiquity, were affordable only by the very wealthy.
Glassblowing. It was probably in the Middle East during the first century B.C.E. that the important technique of glassblowing was discovered. A hollow metal rod (or pipe) was used to pick up a gob of molten glass; the act of blowing into the pipe generated a bubble of glass. If the bubble were blown into a mold, the molten glass could be given a desired shape. Wooden paddles and pincers were used to refine the shape even further. The blowing procedure was used to make glass objects that were larger and thinner than those that had been made previously, and it was much faster than previous glass-forming methods. As glass pieces became easier to make, they became cheaper and more available. The ancient Romans became particularly skillful at glassblowing. More glass was produced and used in the Roman world than in any other civilization of antiquity. During the Middle Ages, there was a great expansion of glassblowing activity, especially in Venice, the Middle East, and European countries such as Spain and Germany.
Some Modern Methods. Since the nineteenth century, many centuriesold glass-forming methods have been mechanized, greatly increasing the rate of production of glass objects, and lowering the prices of these objects. For example, the "ribbon machine," developed in the 1920s for the automatic glassblowing of lightbulbs, is a milestone of mechanical glass forming. In the ribbon machine, puffs of air blow glass bubbles from a rapidly moving ribbon of molten glass into a moving stream of molds that give the bulbs their shape and then release them. Small lightbulb blanks can be made at the rate of 1,000 per minute.
With so many millions of windows in buildings and vehicles everywhere, we tend to take sheets of flat glass for granted. Throughout most of human history, however, there were no sheets of flat, transparent glass. Even as recently as the eighteenth century, glass windows were quite uncommon.
In a very limited way glass windows did start to appear in the Roman world during the third century, but they were generally small glass fragments set in bronze or wooden frames. In that era most windows were not glass, but were thin sheets of translucent horn or marble, or perhaps panes of mica (isinglass). Around 600 C.E. , during the Byzantine period, glass windows (usually made of small pieces of colored glass) began to appear in the large churches, but glass windows in houses and other secular buildings remained quite rare until the end of the eighteenth century.
The principal method for making flat glass during the 1700s called for blowing a hot glass bubble, securing an iron rod to the bubble's other side, and then cutting the bubble free from the blowing pipe. The tulip-shaped hot glass was then rotated rapidly around the iron rod axis until the centrifugal force forced the glass tulip to open up and form a disk. The rod was then removed from the glass (leaving a spot in the middle of the glass disk that looked rather like a bull's eye). This method was the source of the old "bull's-eye" windows that can still sometimes be found in English pubs. The windows were limited in size and poor in optical quality (besides having a bull's-eye at their centers).
The chief method for making flat glass during the 1800s was the cylinder method. The first step was to blow a large glass bubble (compressed air was often used); it would then be swung back and forth until the bubble became elongated and acquired a cylindrical shape; finally the cylinder was split lengthwise, reheated, and allowed to flatten on an iron table. The resulting pane of glass was not really flat, and it had a lot of optical distortion, but the method was used widely to make sheet glass. For example, it was used to produce the 300,000 panes of glass that were used to build London's Crystal Palace, the huge greenhouse constructed for the London World's Fair Exhibition of 1851.
By the twentieth century these methods were replaced by an innovative technique invented by a Belgian named Foucault, who had learned how to draw up continuous sheets from a tank filled with molten glass. Even this glass was of nonuniform thickness and had some roughness at its surface, therefore, for high quality flat glass, it had to be ground and polished.
Then, in 1959, the Pilkington Glass Works in Britain introduced the "float glass" process. In the float process, molten glass is allowed to flow continuously onto a mirrorlike surface of molten tin at 1,000 o C (1,832 o F). At this temperature the glass spreads out and becomes a layer that is about 6 millimeter (1/4 inch) thick. If the layer is stretched as it cools, a thickness of 2 millimeter (0.08 inch) can be achieved. The glass is allowed to advance on the hot liquid tin until, at 600 o C (1,112 o F), it becomes solid enough to be lifted off the molten tin surface. It is then annealed (heated to relieve any strain) before being cut into desired sheet lengths. The float glass method rapidly replaced the Foucault drawing process, and today it is the standard method for making flat glass. A large modern float glass plant can produce 5,000 tons of glass sheet per week, and it can be operated 24 hours a day, 365 days a year, for several years before serious repairs are apt to be needed. Float glass has uniform thickness and bright fire-polished surfaces that need no grinding or polishing.
The drawing of glass fibers had long been of interest, but glass fibers found little use until the twentieth century. Articles such as wedding gowns made from glass fiber cloth were largely curiosities, made for show rather than use. In the 1930s glass researchers learned to feed molten glass into platinum bushings having hundreds of tiny holes. Fine glass filaments of 10 to 50 microns were rapidly drawn downward and assembled as bundles or strands of glass fiber. Today a major use of glass cloth or filaments is to strengthen the plastics used to make fiberglass-reinforced composites. These composites are widely used in making boats, from canoes to yachts, and bodies for cars, such as the Corvette.
An even larger poundage market is that of glass wool insulation. In a process much like that used to make cotton candy, fine glass fibers are spun, sprayed with an organic bonding agent, and then heat-cured and cut into mats of various sizes, to be used for insulating buildings and appliances.
Surely the most significant glass fiber development in recent times is fiber optics, or optical wave guides. These ultrapure, very fine glass fibers are a most crucial part of modern communications technology, wherein glass fibers link telephones, televisions, and computers. A single strand of glass optical fiber that has a protective plastic coating looks much like a human hair. The glass fiber has an inner core of ultrapure fused silica, which is coated with another silica glass that acts as a light-refractive barrier. Lasers are used to convert sound waves and electrical impulses to pulses of light that are sent, static-free, through the inner glass core. Glass fibers can transmit many times more information than can be carried by charges moving in a copper wire. In fact, one pound of glass optical wave guides can transmit as much information as can be transmitted via 200 tons of copper wire. Today millions of miles of optic fibers are crisscrossing not only the United States, but also the entire planet.
Windows need to be cleaned. In 2000 a new glass that largely cleans itself when it comes into contact with rain was introduced. This low-maintenance glass was developed by Pilkington Glass Works, the company that invented the float process. It is made by depositing a microscopically thin coating of titanium dioxide (TiO 2 ) on hot sheet glass during its manufacture in the float process. As dirt collects on the window, the Sun's ultraviolet rays promote a catalytic reaction at the glass surface that breaks down and loosens surface dirt.
Bibliography
Brooks, John A. (1973). Glass. New York: Golden Press.
Douglas, R. W., and Susan Frank (1972). A History of Glassmaking. Oxfordshire, England: G. T. Foulis & Co.
Kampfer, Fritz, and Beyer, Klaus G. (1966). Glass: A World History. London: Studio Vista.
Kolb, Kenneth E., and Kolb, Doris K. (1988). Glass: Its Many Facets. Hillside, NJ: Enslow.
Phillips, Phoebe (1981). The Encyclopedia of Glass. New York: Crown Publishers.
Rogers, Frances, and Beard, Alice (1948). 5000 Years of Glass. New York: Lippincott.
And why they do so.
Do you have a sheet or chart?