Freons
The trademark Freon refers to any of several gaseous chlorofluorocarbons, CFCs, with the general formula CF x Cl 4−x or C 2 F x Cl 6−x . Due to their physical and chemical properties, these CFCs became the compounds of choice as propellants and refrigerants, substituting for the toxic and flammable sulfur dioxide and ammonia materials used until the early 1930s. Due to their long life, Freons drift for years in the atmosphere and eventually find their way to the stratosphere where they decompose and destroy the protective ozone layer. Current legislation calls for the elimination of Freons.
It was at an American Chemical Society (ACS) meeting in 1930 that Thomas Midgley Jr., of General Motors, announced the synthesis of dichlorofluoromethane by the "Swarts process"—a simple and inexpensive technique named after Belgian chemist Frédéric Swarts who worked on fluorine chemistry in the 1890s (Midgley and Henne, 1930, pp. 542–545). Seeking to make two points, Midgley inhaled the vapors and blew out a candle, thus demonstrating that dichlorofluoromethane was neither toxic nor flammable. The E. I. du Pont de Nemours & Co., working with the Frigidaire Division of General Electric in a joint research program called Kinetic Chemicals, registered these CFCs under the trademark of Freons.
Du Pont introduced a system of naming the CFCs according to the numbers of fluorine, hydrogen, and carbon atoms they contain. The number farthest to the right is the number of fluorines; the second digit from the right is the number of hydrogens plus one; and the third digit from the right is the number of carbons minus one. Thus, CHClF 2 is Freon 22; CCl 2 F 2 is Freon 12; CCl 3 F is Freon 11 (the zero, for the number of carbons, does not show); C 2 Cl 2 F 4 is Freon 114; and Freon 113 is C 2 Cl 3 F 3 .
Freons are essentially colorless, odorless, nonflammable, and nontoxic. Their low boiling points, low surface tension, low viscosity, insolubility in water, and general chemical inertness are quite remarkable: They remain stable in hot concentrated mineral acids and are unaffected by molten sodium. These properties result from the strong C–F bonds that become shorter as the fluorine atom to carbon atom ratio increases. Thus, the C–F bond length is 1.39 Å (angstroms) in CH 3 F, 1.358 Å in CF 2 Cl 2 , 1.332 Å in CHF 3 , and 1.323 Å in CF 4 .
Because of their inert properties, Freons were quickly employed as ideal fire extinguishing, cleansing, and foaming agents, as solvents, and as aerosol propellants. Their low-pressure properties allow them to vaporize easily, and thus they were used as refrigerants. While their chemical inertness makes them valuable, it also creates a problem, as they persist in the environment for long periods, eventually diffusing into the stratosphere (altitudes of 25–40 kilometers; 15–25 miles) to decompose and then destroy the protective layer of ozone. This ozone is essential to human and animal life because it absorbs the Sun's ultraviolet light. Without this protection, blindness, genetic damage, and skin cancers can result.
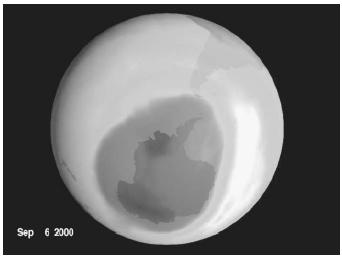
In the mid-1970s F. Sherwood Rowland and M. Molina showed evidence that Freons might drift freely and remain unaffected for up to 100 years, eventually move to the ozone layer, produce free chlorine atoms, and possibly destroy ozone (Molina and Rowland, 1974, pp. 810–812). They thus laid the foundation for a theory of ozone depletion and, together with Paul Crutzen of the Max Planck Institute, received the Nobel Prize for chemistry in 1995. In 1985 Joseph C. Farman, et al. published an article demonstrating the large volume of diminished ozone over Antarctica (Farman, Gardiner, and Shanklin, 1985, pp. 207–210).
In the stratosphere Freons decompose with exposure to ultraviolet light
CCl 2 F 2 (g) + uv light → CF 2 Cl(g) + Cl(g)
and the chlorine atoms destroy ozone
Cl(g) + O 3 (g) → ClO(g) + O 2 (g) O(g) + ClO(g) → Cl(g) + O 2 (g) O(g) + O 3 (g) → 2 O 2 (g)
The oxygen atom is formed in a separate reaction through the light-induced destruction of oxygen molecules.
O 2 + uv light → 2 O
It has been estimated that one chlorine atom has an atmospheric lifetime of one to two years and may destroy 100,000 ozone molecules, thus contributing to the mysterious "ozone hole." The British Antarctic Survey discovered the lowest ozone concentration in Earth's atmosphere ever recorded, in October 1984: nearly 40 percent less than the historical average for Antarctica. Atmospheric currents concentrate CFCs over Antarctica, creating the ozone hole that in 1984 was larger than the United States and taller than Mount Everest. The loss of ozone as a protective layer permits the penetration of increased levels of ultraviolet light to Earth's surface.
Today Freons are banned by an international agreement, and substitutes are sought. The United States has banned the production of CFCs since 1977. Non-ozone-depleting alternatives include hydrofluorocarbons (HFCs) and hydrochlorofluorocarbons (HCFCs) such as CH 2 FCF 3 (HFC 134a) and CHCl 2 CF 3 (HCFC 123). In 1987 an international treaty, the Montreal Protocol, called for reducing CFCs, and a 1992 amendment to the treaty called for an end to CFC production. By 1993 CFC emissions had dropped dramatically. A total of 148 countries have now signed the Montreal Protocol calling for HCFCs to be phased out by 2020 and replaced by HFCs, which contain no chlorine and have a short lifetime. The black market of CFCs is of such proportion that Scientific American has reported (Beardsley, 1998, p. 32) that "the illegal CFC trade is one of the greatest threats to ozone-layer recovery."
SEE ALSO Organic Halogen Compounds ; Ozone .
Erwin Boschmann
Bibliography
Beardsley, Tim (1998). "Hot Coolants." Scientific American 279(1):32.
Cogan, Douglas G. (1988). Stones in a Glass House: CFCs and Ozone Depletion. Washington, DC: Investor Responsibility Research Center.
Elkins, James W. (1999). "Chlorofluorocarbons (CFCs)." In The Chapman & Hall Encyclopedia of Environmental Science, ed. David E. Alexander and Rhodes W. Fairbridge. Boston: Kluwer Academic.
Farman, Joseph C.; Gardiner, Brian G.; and Shanklin, J. D. (1985). "Large Losses of Total Ozone in Antarctica Reveal Seasonal ClO x /NO x Interaction." Nature 315: 207–210.
Hileman, Bette (2002). "Nations Fight CFC Smuggling." Chemical and Engineering News 80(12):30–32.
Midgley, Thomas, Jr., and Henne, Albert L. (1930). "Organic Fluorides as Refrigerants." Industrial and Engineering Chemistry 22:542–545.
Miller, G. Tyler, Jr. (2000). "Global Warming and Ozone Loss." In Living in the Environment: Principles, Connections, and Solutions, 11th edition. Pacific Grove: Brooks/Cole.
Molina, M. J., and Rowland, F. Sherwood (1974). "Stratospheric Sink for Chlorofluoromethanes: Chlorine Atom Catalyzed Destruction of Ozone." Nature 249: 810–812.
Internet Resources
Montréal Protocol on Substances that Deplete the Ozone Layer. Amended in London, June 1990; and in Copenhagen, November 23–25, 1992. Available from http://www.tufts.edu/departments/ .
The key to the code is to add 90 to the number; the result shows the number of C, H, and F atoms. For HCFC-141b:
141 + 90 = 231
#C #H #F
One more piece of information is needed to decipher the number of Cl atoms. All of these chemicals are saturated; that is, they contain only single bonds. The number of bonds available in a carbon-based molecule is 2C + 2. Thus, for HCFC-141b, which has 2 carbon atoms, there are 6 bonds. Cl atoms occupy bonds remaining after the F and H atoms. So HCFC-141b has 2C, 3H, 1F, and 2Cl:
HCFC-141b = C 2H 3FCl 2
Notice that the HCFC designation ( h ydro c hloro f luoro c arbon) is a good double-check on the decoding; this molecule does, indeed, contain H, Cl, F, and C. The "b" at the end describes the isomer.
Ex #2: HFC-134a.
134 + 90 = 224
#C #H #F
Again, there are 6 bonds. But in this case, there are no bonds left over after F and H, so there are no chlorine atoms. Thus:
HFC-134a = C 2H 2F 4
In this case, too, the prefix is accurate: this is an HFC ( h ydro f luoro c arbon), so it contains only H, F, and C, but no chlorine.
One final example: PFC-218.
218 + 90 = 308
#C #H #F
This time, there are 2 x 3 + 2 = 8 bonds. However, there are no bonds left over after F, so there are no chlorine atoms or H atoms. Thus:
PFC-218 = C 3F 8
again, the prefix is accurate: this is a PFC ( p er f luoro c arbon), so it contains only F, and C.
Any molecule with only 1C (e.g., CFC-12) will have a 2-digit number, while those with 2C or 3C will have a 3-digit number.
Maybe it's a different route to the same destination...