Coenzyme
Coenzymes are small organic molecules that link to enzymes and whose presence is essential to the activity of those enzymes. Coenzymes belong to the larger group called cofactors, which also includes metal ions; cofactor is the more general term for small molecules required for the activity of their associated enzymes. The relationship between these two terms is as follows
I. Cofactors
- Essential ions
- Loosely bound (forming metal-activated enzymes)
- Tightly bound (forming metalloenzymes
- Coenzymes
- Tightly bound prosthetic groups
- 2 Loosely bound cosubstrates
Many coenzymes are derived from vitamins . Table 1 lists vitamins, the coenzymes derived from them, the type of reactions in which they participate, and the class of coenzyme.
Prosthetic groups are tightly bound to enzymes and participate in the catalytic cycles of enzymes. Like any catalyst , an enzyme–prosthetic group complex undergoes changes during the reaction, but before it can catalyze another reaction, it must return to its original state.
Flavin adenine dinucleotide (FAD) is a prosthetic group that participates in several intracellular oxidation -reduction reactions. During the catalytic cycle of the enzyme succinate dehydrogenase, FAD accepts two electrons from succinate, yielding fumarate as a product. Because FAD is tightly bound to the enzyme, the reaction is sometimes shown this way
succinate + E–FAD → fumarate + E–FADH 2
where E–FAD stands for the enzyme tightly bound to the FAD prosthetic group. In this reaction the coenzyme FAD is reduced to FADH 2 and remains tightly bound to the enzyme throughout. Before the enzyme can catalyze the oxidation of another succinate molecule, the two electrons now belonging to E–FADH 2 must be transferred to another electron acceptor, ubiquinone. The regenerated E–FAD complex can then oxidize another succinate molecule.
Cosubstrates are loosely bound coenzymes that are required in stoichiometric amounts by enzymes. The molecule nicotinamide adenine dinucleotide (NAD) acts as a cosubstrate in the oxidation-reduction reaction that is catalyzed by malate dehydrogenase, one of the enzymes of the citric acid cycle.
malate + NAD + → oxaloacetate + NADH
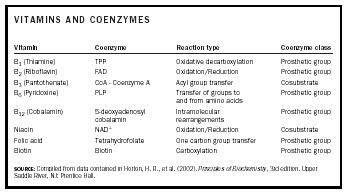
| VITAMINS AND COENZYMES | |||
| Vitamin | Coenzyme | Reaction type | Coenzyme class |
| SOURCE: Compiled from data contained in Horton, H. R., et al. (2002). Principles of Biochemistry , 3rd edition. Upper Saddle River, NJ: Prentice Hall. | |||
| B 1 (Thiamine) | TPP | Oxidative decarboxylation | Prosthetic group |
| B 2 (Riboflavin) | FAD | Oxidation/Reduction | Prosthetic group |
| B 3 (Pantothenate) | CoA - Coenzyme A | Acyl group transfer | Cosubstrate |
| B 6 (Pyridoxine) | PLP | Transfer of groups to and from amino acids | Prosthetic group |
| B 12 (Cobalamin) | 5-deoxyadenosyl cobalamin | Intramolecular rearrangements | Prosthetic group |
| Niacin | NAD + | Oxidation/Reduction | Cosubstrate |
| Folic acid | Tetrahydrofolate | One carbon group transfer | Prosthetic group |
| Biotin | Biotin | Carboxylation | Prosthetic group |
In this reaction, malate and NAD + diffuse into the active site of malate dehydrogenase. Here NAD + accepts two electrons from malate; oxaloacetate and NADH then diffuse out of the active site. The reduced NADH must then be returned to its NAD + form. For each catalytic cycle, a "new" NAD + molecule is needed if the reaction is to occur; thus, stoichiometric quantities of the cosubstrate are needed. The reduced form of this coenzyme (NADH) is converted back to the oxidized form (NAD + ) via a number of simultaneously occurring processes in the cell, and the regenerated NAD + can then participate in another round of catalysis.
Coenzymes, then, are a type of cofactor. They are small organic molecules that bind tightly (prosthetic groups) or loosely (cosubstrates) to enzymes as they participate in catalysis.
SEE ALSO Active Site ; Cofactor ; Enzymes .
Paul A. Craig
Bibliography
Berg, Jeremy M.; Tymoczko, John L.; and Stryer, Lubert (2002). Biochemistry , 5th edition. New York: W. H. Freeman.
Horton, H. Robert; Moran, Laurence A.; Ochs, Raymond S.; Rawn, David J.; and Scrimgeour, K. Gray (2002). Principles of Biochemistry , 3rd edition. Upper Saddle River, NJ: Prentice Hall.
Voet, Donald; Voet, Judith G.; and Pratt, Charlotte (1999). Fundamentals of Biochemistry. New York: Wiley.
Comment about this article, ask questions, or add new information about this topic: