Fibrous Protein

Many of the familiar cells in your body use fibrous proteins to carry out important tasks. Skin, bones, muscles, tendons, and hair cells all rely on molecules in this class.
The amino acid sequences of fibrous proteins often contain repeating sets of amino acid residues . Such repeating sets tend to cause the protein to be both elongated and strong.
Human hair provides a good example of how fibrous proteins have structural functions. The main protein in hair is called alpha-keratin. More than twenty-five variations of alpha-keratin are found in the cells of mammals. Individual molecules of alpha-keratin are not particularly large, but they provide the structure for hair by forming a coil of coils. Clustering several smaller structures to make a larger filament adds strength in the same way that strong ropes are made from groups of smaller twine.
Fibrous proteins also gain strength due to interactions between the side chains of the residues. The alpha-keratin polypeptides , for example, have a large number of cystine residues, which can form disulfide bonds .
When a person gets a "permanent," the first chemicals that are applied break the disulfide bonds. The hair is then curled (or straightened), and
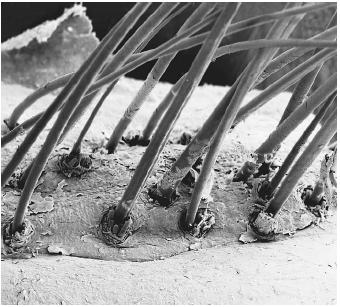
another chemical is applied to reform the disulfide bonds so the new style is retained permanently.
Coils such as those found in alpha-keratin are not the only structural motifs present in fibrous proteins. Silk, for example, is largely composed of fibrous proteins whose structures resemble interleaved sheets.
SEE ALSO Quaternary Structure ; Secondary Structure ; Tertiary Structure .
Thomas A. Holme
Bibliography
Branden, Carl, and Tooze, John (1991). Introduction to Protein Structure, 2nd edition. New York: Garland Publishing.
Kaplan, David; Adams, W. W.; Farmer, B.; and Vinney, C., eds. (1994). Silk Polymers: Materials Science and Biotechnology. Washington, DC: American Chemical Society.
Voet, Donald, and Voet, Judith G. (1995). Biochemistry, 2nd edition. New York: Wiley.
Comment about this article, ask questions, or add new information about this topic: