Double Helix
Described in 1953 by James Watson and Francis Crick, the double helix of DNA (deoxyribonucleic acid) is the cellular storehouse of genetic information. This biopolymer consists of a pair of complementary chains approximately 2.4 nanometers (9.5×10 −8 inches) in diameter and composed of
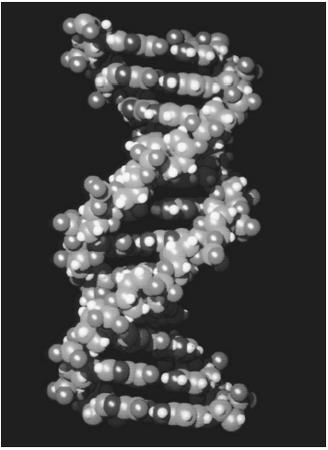
deoxyribose sugar molecules linked to each other by phosphoric acid, connecting the number three carbon of one sugar to the number five carbon of another. Attached to each sugar is a heterocyclic base: adenine , guanine , cytosine , or thymine . Each turn of the helix contains about 10.4 nucleotides.
These chains are said to be complementary: Guanine on one chain always pairs with cytosine on the opposite strand of DNA by forming three hydrogen bonds , and adenine on one chain pairs with thymine on the complementary chain, held in position by two hydrogen bonds. The interchain bonding forms an attraction between the two DNA chains and stabilizes the double helix against the strong repulsive force of the phosphoric acid residues. The chains are said to be antiparallel; that is, the two chains are held closely together but run in opposite directions, with the 3′ end of one chain matching the 5′ end of the other chain.
DNA performs two important functions. It contains the genetic code that provides directions for replication ( synthesis of new DNA), and thus serves as a storehouse of genetic information, allowing the physical characteristics of parent organisms to be passed on to offspring. DNA also acts as a storehouse of synthetic information. The individual genes can be turned on or off, allowing the information of each codon within a gene to be converted into information contained in messenger RNA (a process known as transcription ). This information can then be transcribed into protein having catalytic or structural properties.
CHARGAFF'S RULES
Erwin Chargaff found that the ratios of adenine to thymine and of guanine to cytosine were always 1:1, suggesting that these bases form pairs. The fact that the ratios are 1:1 is referred to as Chargaff's rules.
SEE ALSO Base Pairing ; Codon ; DNA Replication ; Hydrogen ; Nucleic Acids ; Watson, James Dewey .
Dan M. Sullivan
Bibliography
Boyer, Rodney F. (2002). Concepts in Biochemistry. New York: Wiley.
Devlin, Thomas M., ed. (2002). Textbook of Biochemistry: With Clinical Correlations, 5th edition. New York: Wiley-Liss.
McKee, Trudy, and McKee, James R. (2003). Biochemistry: The Molecular Basis of Life, 3rd edition. Boston: McGraw-Hill.
Comment about this article, ask questions, or add new information about this topic: