Terpenes

If you walk into a garden in bloom and breathe deeply, you are likely to encounter great smells. In many cases, the molecules that bring those scents to your nose are terpenes. Terpenes are a class of molecules that typically contain either ten or fifteen carbon atoms built from a five-carbon building block called isoprene . It is important to note these molecules are volatile —they tend to evaporate relatively easily—which allows them to reach one's nose and trigger the olfactory senses. Figure 1 shows the structure of several molecules in this category and identifies the plant from which they are derived.
Because they share the common building block of isoprene, terpenes can be categorized based on how many terpene units they include. The "base" of this system is a two-isoprene unit, so monoterpenes have ten carbons. This category includes molecules such as camphor, menthol, and pinene. If three isoprenes are present, as in farnesol (shown in Figure 1), the category is called sesquiterpenes.
In plants, terpenes are typically found as a component of the essential oils. The name of this fluid suggests an important characteristic of this class of molecules. If humans have found these oils important enough to refer to them as essential, there is a reasonable chance that they have useful properties. Many terpenes such as menthol (shown in Figure 1) and camphor have medicinal values. You may have used a throat lozenge with menthol to help clear your sinuses the last time you had a cold.
Another use of terpenes may be found in coatings such as varnish. Varnish for musical instruments like violins can be made largely from the terpene pinene. The key to generating a coating such as varnish is to allow polymerization to occur. The fact that terpenes are built up from isoprene building blocks, much like any other polymer, suggests that polymerization is a possibility. When pinene (present in pine tree and made into turpentine) is exposed to air and sunlight, it will slowly polymerize and make a fine finish for wood.
β- carotene is a tetraterpene (structure shown in Figure 1); it has four isoprene units. This molecule, found in abundance in carrots, is useful in
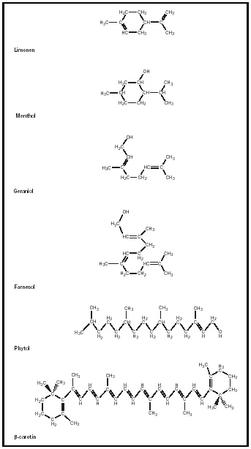
human diets because it can be converted into vitamin A in the body. It also plays an important role in plants because it is among the pigments that absorb light in photosynthesis . When leaves change color in the fall, β- carotene is often responsible for this effect.
Bibliography
Gimelli, Salvatore Paul (2001). Aroma Science. Port Washington, NY: Micelle Press.
Maarse, Henk, ed. (1991). Volatile Compounds in Foods and Beverages. New York: Marcel Dekker.
Comment about this article, ask questions, or add new information about this topic: