Dimitri Mendeleev

RUSSIAN CHEMIST
1834–1907
Dimitri Ivanovich Mendeleev (or Mendeleyev or Mendelejeff) was born in Tobolsk, Siberia, on January 27, 1834. He was the fourteenth and youngest child of the family. His father was the director of the Tobolsk Gymnasium (high school). Tragedy plagued the family in Mendeleev's early years. His father became blind and was forced to retire from his job, and then unexpectedly died. His mother supported the family by managing a glass factory, but in 1848 it burned to the ground. His mother moved the family first to Moscow and then to St. Petersburg. In 1850 Mendeleev began his training as a teacher, following in his father's footsteps at the Pedagogical Institute in St. Petersburg. A few months after this, his mother and older sister died of tuberculosis.
When Mendeleev graduated, he moved to Simferopol on the Crimean Peninsula to assume a post as a science teacher, but the school was soon closed because of the Crimean War. He returned to St. Petersburg and received a master's degree in 1856 after presenting his thesis "Research and Theories on Expansion of Substances Due to Heat."
The years 1859 to 1861, when the Ministry of Public Instruction sent him abroad to study, shaped Mendeleev's career as a scientist. He studied gas density with the chemist Henri Victor Regnault in Paris and spectroscopy with the physicist Gustav Kirchhoff in Heidelberg. It was while working in Heidelberg that Mendeleev discovered the principle of critical temperature for gases. Once a gas is heated to a temperature above its critical point, no amount of pressure will turn it into a liquid. His work went unnoticed, and the discovery of critical temperatures is usually attributed to the Irish physicist and chemist Thomas Andrews.
Mendeleev also attended the 1860 Karlsruhe Congress, the first international chemistry conference. Many of the leading chemists of the day were in attendance, and one of the central questions addressed was the appropriate method for calculating atomic weight . Different chemists used different systems, leading to widespread confusion over everything from nomenclature to chemical formulas. Mendeleev heard the Italian chemist Stanislao Cannizzaro present Amedeo Avogadro's hypothesis that equal volumes of gas under equal temperature and pressure contained equal numbers of molecules.
Mendeleev returned to St. Petersburg determined to make a name for himself and build on the innovations to which he had been exposed. He became a professor of chemistry at the Technological Institute in 1863. His attention to science also extended to practical application, and he often worked as a consultant to the government on farming, mining, and oil production.
In 1866 Mendeleev became professor of general chemistry at the University of St. Petersburg. Finding that no modern organic chemistry textbook existed in Russian, Mendeleev decided to write one (it became a classic work, going through many editions). It was in the course of this project that he made his most important contribution to chemistry. Principles of
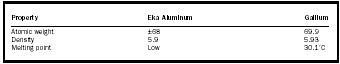
| Property | Eka Aluminum | Gallium |
| Atomic weight | ±68 | 69.9 |
| Density | 5.9 | 5.93 |
| Melting point | Low | 30.1°C |
Chemistry was not a mere compilation of facts; it presented chemistry as a unified study. At its heart was the relationship of the elements.
On February 14, 1869, Mendeleev began work on the chapter that would discuss the elements. He already believed that there was some underlying principle connecting the elements. He transcribed his notes onto a set of cards, one for each element containing everything he knew about that element. He arranged and rearranged the cards until he was struck by a similarity between his arrangements and those of the card game patience (solitaire), in which cards are sorted by suit and then in descending numerical order. Exhausted, Mendeleev fell asleep. When he awoke, he devised a grouping of the elements by common property in ascending order of atomic weight. He called his innovation the Periodic Table of the Elements.
Within weeks, Mendeleev's Periodic Table was presented to the Russian Chemical Society and was published in the Journal of Russian Physical Chemistry ; it was published later the same year in the prestigious German journal Zeitschrift für Chemie. Revised and expanded tables appeared in the Annalen der Chemie in 1872. Since the German journals were known to every research chemist, Mendeleev's Periodic Table became widely known almost at once. Although details of the tables were subject to argument, and many newly discovered elements were later added, the basic principle of organization behind the table was quickly accepted.
The true insight that informed Mendeleev's work was shown not just in what he had included in the Periodic Table, but also in what he had left out. He did not assume that all elements were known. Where there was a significant gap in atomic weights between the elements in the table, he left a gap in the table. He posited that there were undiscovered elements that existed in the gaps and even predicted the characteristics of three of them. He called these eka boron, eka aluminum, and eka silicon ( eka being Sanskrit for "first"). See Tables 1 through 3 for the properties of these elements.
When these elements were eventually discovered, and because his system agreed with one developed independently by the German chemist Lothar Meyer in 1864, Mendeleev achieved widespread fame. The Periodic Table of the Elements provided a unifying system for classifying and understanding the elements and their function in the composition of matter.
Mendeleev received the Davy Medal (with Meyer in 1882) and the Copley Medal (in 1905), but Russia's Imperial Academy of Sciences refused to acknowledge his work. He resigned his university position in 1890 and was

| Property | Eka Boron | Scandium |
| Atomic weight | 44 | 44.1 |
| Density of oxide | 3.5 | 3.8 |
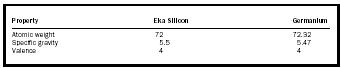
| Property | Eka Silicon | Germanium |
| Atomic weight | 72 | 72.32 |
| Specific gravity | 5.5 | 5.47 |
| Valence | 4 | 4 |
appointed director of the Bureau of Weights and Measures, holding this job until his death on January 20, 1907.
SEE ALSO Avogadro, Amedeo ; Cannizzaro, Stanislao ; Meyer, Lothar ; Periodic Table .
Andrew Ede
Bibliography
Kelman, Peter, and Stone, Harris (1970). Mendeleyev: Prophet of Chemical Elements. Englewood Cliffs, NJ: Prentice-Hall.
Mendeleyev, Dmitry Ivanovich (1969). Principles of Chemistry. New York: Kraus Reprint.
Strathern, Paul (2000). Mendeleyev's Dream. New York: Berkley Publishing Group.
Internet Resources
ChemNet (Chemistry Department, Moscow State University). "Dmitriy Mendeleev Online." Available from http://www.chem.msu.su/eng/misc/mendeleev .
Comment about this article, ask questions, or add new information about this topic: