Ibuprofen
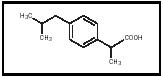
Ibuprofen is a well-known drug that possesses analgesic (pain-relieving) and antipyretic (fever-reducing) properties. It is particularly known for its use in pain relief from arthritis. Ibuprofen was discovered by Dr. Stewart Adams and his colleagues in the United Kingdom in the 1950s, patented in 1961, and first made available in 1969. It became available in the United States in 1974. Ibuprofen tablets are sold under the trade names Advil and Motrin. The chemical name of ibuprofen is 2-(4-isobutylphenyl)propanoic acid.
Ibuprofen also has anti-inflammatory properties, and it belongs to a class of therapeutic agents known as nonsteroidal anti-inflammatory drugs, or NSAIDs. Like acetylsalicylic acid (aspirin), another NSAID, and acetaminophen, ibuprofen works by inhibiting the activity of a class of enzymes called cyclooxygenases (COX). These enzymes are significant because they catalyze the synthesis of prostaglandins, molecules that have both positive and negative effects in the body. Prostaglandins are, for example, protective against the development of stomach ulcers, but they can also mediate inflammation (as well as the pain response).
One of the most interesting things about human COX enzymes is that there is more than one of them—definitely two, and probably at least three. This is important to our understanding of the therapeutic effects of ibuprofen, aspirin, and acetaminophen. It had long been suspected that there was more than one COX enzyme, but it was not until 1991 that evidence for the existence of two forms, COX-1 and COX-2, materialized. It was then recognized that COX-1 is present at near constant levels in the body under all conditions (that is, it is a constitutive enzyme), whereas the levels of COX-2 could increase in response to inflammatory conditions (i.e., it is an inducible enzyme). This led to the idea that the side effects of ibuprofen and aspirin (including stomach ulcers) probably arose from inhibition of the constitutive COX-1 enzyme, whereas the therapeutic benefits arose from inhibition of the inducible COX-2 enzyme.
Ibuprofen and aspirin both inhibit COX-1 and COX-2, but they do it in different ways. Ibuprofen binds noncovalently to a COX enzyme and thus competes with the enzyme's natural substrate. (This is referred to as reversible
inhibition.) On the other hand, aspirin forms a covalent bond to a serine residue in the enzyme, and this bond cannot be broken. (This is called irreversible inhibition.) Acetaminophen does not interact with either COX-1 or COX-2, but it is now understood that it may interact with a newly identified cyclooxygenase, COX-3. Selective targeting of the COX enzymes is an exciting area of pharmacology, in which the challenge continues to be the development of drugs that interact with specific COX enzymes.
SEE ALSO Acetylsalicylic Acid ; Acetaminophen ; Pharmaceutical Chemistry .
Ian S. Haworth
Bibliography
Flower, Rod J. (2003). "The Development of Cox 2 Inhibitors." Nature Reviews Drug Discovery 2(3):179–191.
Internet Resources
International Ibuprofen Foundation. "The History of Ibuprofen." Available from http://www.ibuprofen-foundation.com/history.htm .
RxList. "Ibuprofen." Available from http://www.rxlist.com .