Hair Dyes and Hair Treatments
Hair has no vital function in the human body but provides an outward sign of health and social communication. The history of hair coloring dates from ancient dynasties of Egypt and China where mineral and plant dyes were widely available to cosmeticians. Through the ages, women in particular have dyed their hair to hide grayness and to improve their appearance.
General Features of Hair
In all mammals, hair develops as an epidermal structure from papillae deep in the skin and acquires characteristic patterns on the scalp, eyebrows, eyelashes, and elsewhere on the body. In humans, hair growth is continuous throughout life (declining with advancing age), occurs in cyclic patterns, and
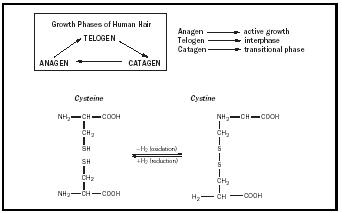
is influenced by androgens , thyroid hormones, and dietary factors. Human hair is composed largely of keratin and consists of a narrow central medulla surrounded by a thick envelope (cortex) of elongate cells, which contain numerous melanin granules that determine the natural color. The hair is ensheathed in a multilayered cuticle of overlapping cells that become progressively imbricated (having edges overlapping in a regular arrangement) with continued growth. These cuticular cells are rich in cystine ( disulfide bonds ) and become rough or show a weathered appearance through exposure to environmental exposure or poor health.
Chemically, human hair contains approximately 85 percent protein, 7 percent water, 3 percent lipid , 4.7 percent protein-bound sulfur (as cystine), and low concentrations of trace minerals (e.g., iron, zinc, copper). The phosphorus content is approximately 80 milligrams per 100 grams (0.003 ounces per 3.5 ounces) of hair. Hair is normally associated with sebum and exocrine secretions from skin glands that confer greasiness but influence its water content and mechanical and physical properties.
Hair follicles are determined prenatally; about 100,000 hairs are found on the scalp region of most adults. Hair density, color, and condition vary according to age, race, and genetic background. Natural hair colors vary from albino or white to blond, red, or intense black and reflect ethnic origin (Caucasoid, Negroid, etc.), age, diet, and health. While hair color is closely related to the density of melanin granules, impairment in a person's health or substances in the diet that influence the availability of trace minerals are potential causes of changes in hair color or condition. The configuration of the hair shaft (i.e., straight, wavy, spiral, or peppercorn) is attributable to the number and distribution of disulfide bonds. Hair straightening requires reduction of these disulfide bonds and fiber cross-linkages.
Hair serves to eliminate toxic materials (e.g., lead) and metabolites from the body, and may be used to monitor environmental contamination. For example, copper deficiency is a cause of Menke's "kinky" hair syndrome; protein deficiency leads to hair loss and discoloration. Hair keratin carries a strong negative charge and binds inorganic materials; it becomes prone to discoloration through exposure to environmental chemicals (e.g., cobalt, tar in cigarettes, picric acid, trinitrotoluene, etc.). Prolonged exposure to copper in diet, tap water, or swimming pools is a cause of green hair.
Natural Hair Color and Its Control
Melanin granules are secreted by melanocytes in the hair papilla and distributed to keratin in the hair cortex and inner layers of the hair sheath during normal development. Melanogenesis is subject to hormonal control and has been the focus of intensive genetic studies. Two main forms of melanin exist in human skin—eumelanin and phaeomelanin, both of which are derived from tyrosine through the action of tyrosinase (a cupro-enzyme) and possibly other key enzymes (with nickel, chromium, iron, and manganese as cofactors). Tyrosine is converted to dihydroxyphenylalanine and, via a series of intermediate steps, to indole-5,6-quinone, which polymerizes to eumelanin. Phaeomelanins are produced by a similar mechanism but with the incorporation of sulfur (as cysteine) by a nonenzymatic step in the oxidation process.
Hair color is a balance between these two melanins. Albino or white-haired individuals have latent melanocytes, but possibly show defects in tyrosinasemediated events. Graying of the hair is age-related and possibly results from declining melanocytic function or retarded hair growth resulting from atrophy or degenerative changes in hair papillae. Hair melanin absorbs insufficient ultraviolet (UV) light energy to afford protection for most individuals against sunburn.
Hair Dyes and Cosmetic Coloring
A wide variety of dyes, dressings, and conditioners are available to men and women to enhance the color of hair or to alter its condition, providing the "feel good" factor. Natural hair dyes such as henna and mineral salts are still used, but hair dyeing increasingly involves careful chemical manipulation of the chemistry of hair fibers through bleaching or enhancement of natural colors. Additionally, social and cultural customs have led to the increasing demand for exotic colors (reds, greens, blues, yellows, etc.). Hair coloring is a well-defined science with intense study of the interaction between hair keratin and highly reactive organic dyes, oxidizing agents, and conditioners.
Available hair dyes include:
- Minerals such as lead acetate (<1% aqueous), lead sulfide (kohl); silver nitrate; salts of bismuth, copper, and cobalt (commonly called "gradual" colorants)
- Vegetable materials such as henna (flowers and leaves of Lawsonia inermis that contain acidic naphthoquinone, chamomile, and indigo)
- Synthetic dyes including a large number of organic dyes to provide permanent, semipermanent, temporary, or progressive color changes or to enhance natural colors
The dyeing process provides for temporary, semipermanent (direct dyes), and oxidation-type reactions (semipermanent or permanent colors). It may involve absorption or adsorption (electrostatic) of the colorant into/to the hair structure, bleaching or otherwise masking the natural melanin colors,
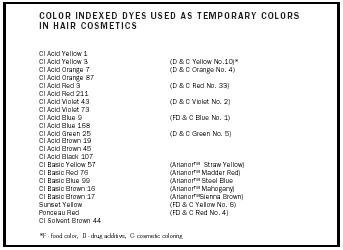
| COLOR INDEXED DYES USED AS TEMPORARY COLORS IN HAIR COSMETICS | |
| *F - food color, D - drug additive, C- cosmetic coloring | |
| CI Acid Yellow 1 | |
| CI Acid Yellow 3 | (D & C Yellow No.10)* |
| CI Acid Orange 7 | (D & C Orange No. 4) |
| CI Acid Orange 87 | |
| CI Acid Red 3 | (D & C Red No. 33) |
| CI Acid Red 211 | |
| CI Acid Violet 43 | (D & C Violet No. 2) |
| CI Acid Violet 73 | |
| CI Acid Blue 9 | (FD & C Blue No. 1) |
| CI Acid Blue 168 | |
| CI Acid Green 25 | (D & C Green No. 5) |
| CI Acid Brown 19 | |
| CI Acid Brown 45 | |
| CI Acid Black 107 | |
| CI Basic Yellow 57 | (Arianor™ Straw Yellow) |
| CI Basic Red 76 | (Arianor™ Madder Red) |
| CI Basic Blue 99 | (Arianor™ Steel Blue |
| CI Basic Brown 16 | (Arianor™ Mahogany) |
| CI Basic Brown 17 | (Arianor™Sienna Brown) |
| Sunset Yellow | (FD & C Yellow No. 6) |
| Ponceau Red | (FD & C Red No. 4) |
| CI Solvent Brown 44 | |
or alteration of the structure of the hair shaft, allowing deep penetration of the colorant. The hair cuticle provides a barrier to the absorption of hair dyes, particularly those of high molecular weight, but damaged fibers exhibiting higher negative charges and reduced phospholipid content are more porous. Dye uptake is determined by the partition coefficient between the hair and the dye carrier (water, alcohol, etc.), pH, and chemical charge (dye-fiber interaction). Thickeners and surfactants can also influence dye uptake.
The vast array of natural dyestuffs, metallic salts, and synthetic organic chemicals available to cosmeticians are regulated in the United States (by the Food and Drug Administration [FDA]), the European Union, and many other countries, and regulatory toxicity information is documented.
Henna. Henna is the oldest and most widely used vegetable dye utilized in hair coloring. A temporary chestnut color is produced in blond or auburn hair by applying a paste of henna flowers and leaves ground in hot water immediately before use. (The dye is unstable in aqueous solution .) The addition of indigo achieves darker blue-black shades; extracts of walnut shell or logwood enhance brown coloration.
Mineral dyes. Of the mineral dyes, only lead acetate is commercially available. In the United States, the FDA permits maximal concentrations of 0.6

| NATURAL DYES FROM PLANTS USED AS HAIR COLORANTS | |
| Plant | Hair Colorant |
| Henna ( Lawsonia inermis ) | 2-hydroxy-1,4-naphthaquinone |
| Chamomile | 4′,5,7trihydroxyflavone |
| Indigo ( Indogofera sp.) | CI Pigment Blue 66 - C 16 H 10 N 2 O 2 |
| Logwood - haematein | C 16 H 12 O 6 |

percent; in the United Kingdom, less than 1 percent is allowed. (Kohl or "suma," an eyelash and eyelid cosmetic used in Asian countries, contains up to 80 percent lead and is a known cause of anemia.) In contact with hair, the lead salt is poorly absorbed but interacts with keratin to deposit lead sulfide (at permitted levels [<0.5%] lead is absorbed into the skin without toxic implications). Silver nitrate has been used to color hair brown-black without significant absorption of metal into the circulation; silver is deposited in the hair cortex as silver sulfide. Metallic hair dyes tend to be long-lasting and are lost as hair grows and is shed naturally. Hair coloration is achieved as a gradual process through repeated application of rinses or pastes over several days.
Temporary dyes. Temporary dyes are frequently acidic and provide short-term coloration (i.e., wash out after one shampoo). They are of low penetration and do not involve melanin bleaching or structural changes in the hair fiber. They are commonly identified with a color index (CI) number or as U.S. Certified Colors; they are water-soluble, high-molecular-weight pigments. Temporary dyes are employed in water-thin color rinses, colored hair-setting lotions, colored styling gels, lotions, and shampoos. Hair dyeing is improved when the colors are applied under moderate heat. Hair dyeing and color balance is difficult to control with temporary methods because of the inconsistency of dye delivery systems and instability of the dye-keratin "bonding."
ESSENTIAL INGREDIENTS OF OXIDATIVE PERMANENT AND SEMIPERMANENT DYEING SYSTEMS
- Surfactant
- Solvent
- Alkalizing Agent
- Oxidant
- Dye
Semipermanent dyes. Semipermanent dyeing systems are designed to last for several weeks. They impart darker colors without the use of oxidizing agents or structural changes in the hair shaft or papillae. Melanin may be partly masked but is not bleached. These dyes diffuse more readily into and out of the hair (aided by solvents and surfactants) and are progressively lost with hair shampooing and normal growth. The method employs dyes that
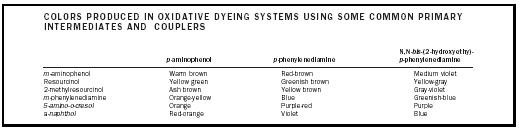
| COLORS PRODUCED IN OXIDATIVE DYEING SYSTEMS USING SOME COMMON PRIMARY INTERMEDIATES AND COUPLERS | |||
| p -aminophenol | p -phenylenediamine | N,N- bis -(2-hydroxyethy)- p -phenylenediamine | |
| m -aminophenol | Warm brown | Red-brown | Medium violet |
| Resourcinol | Yellow green | Greenish brown | Yellow-gray |
| 2-methylresourcinol | Ash brown | Yellow brown | Gray-violet |
| m -phenylenediamine | Orange-yellow | Blue | Greenish-blue |
| 5-amino-o-cresol | Orange | Purple-red | Purple |
| a- naphthol | Red-orange | Violet | Blue |
are already colored, and in general is without the toxic risks associated with semipermanent or permanent methods, which involve de novo color production and strong oxidizing agents. When the dye is lost through shampooing, the hair resumes its natural color.
Oxidative dyes. Oxidative hair dyeing systems involve the use of more toxic reagents . They are multistep processes leading to semipermanent or permanent coloration, according to the extent of bleaching involved. Surfactants and solvents influence the penetration of the active constituents; alkalizing agents determine pH. The resulting hair colorations are more stable against normal wearing processes than semi permanent preparations and involve an initial oxidation reaction, a coupling reaction, and production of a color reaction with dyeing of the hair fiber. The process requires a primary intermediate, a m -coupler or secondary intermediate (color modifiers), and hydrogen peroxide.
Hydrogen peroxide is commonly used as the oxidizing agent; it has the capacity to bleach melanin but it initiates the first coupling reaction and the ultimate development of the color. Initial oxidation of primary intermediates (e.g., p -aminophenol, p -phenylenediamine) by hydrogen peroxide is followed by coupling with an agent like resourcinol, phenols, m -aminophenols, or m -phenylenediamines. Further oxidation of this secondary intermediate leads to the formation of colored indamines, indolanilines, and indophenols. As a general rule, the higher the electron-donating capacity of the coupling agent (especially unsubstituted carbocyclic m -couplers), the higher the absorbance maximum of the indo-dye formed. In the presence of couplers, diand monoimines react to produce indo-dyes.
Many of the organic aromatic amines used in hair dyes are strong sensitizers and oxidative dyes should be used with extreme caution. P -phenylenediamine and its derivatives, commonly employed in permanent or semipermanent hair colorings, are strong sensitizing agents and may damage the hair. Although a large number of possible combinations of primary intermediates and couplers leading to the production of exotic hair colors is possible, the cost of conducting regulatory toxicological evaluation is prohibitive in developing many interesting colors.
Bleaches
Lightening or removal of hair color without structurally damaging the hair shaft is a difficult process. Oxidizing agents, including hydrogen peroxide, can be extremely harmful to hair and will oxidize some cystine to cysteic acid, rendering the fibers less cohesive and susceptible to hydration and swelling under alkaline conditions. Bleached hair shows a loss of melanin granules at the periphery of the hair. Hair color is lost (platinum blond appearance) and the fibers become dry and fragile. Bleaching is inhibited by shampoos or acid rinses.
Hair Conditioners
Hair conditioning includes permanent waving, straightening, and setting. In each case, sequential chemical treatments lead to modifications of the hair shaft with temporary or prolonged changes in disulfide bond distribution and behavioral characteristics. Softening, reshaping, and hardening are integral to permanent waving and involve an initial reduction in disulfide bonds and adhesiveness of adjacent hairs using heat or steam, thioglycollates, or ammonium hydroxide; styling with heated rollers; and setting or neutralizing the reaction. This last event is a reversal of the earlier reduction process with reconstitution of disulfide bonds from adjacent cystine moieties using hydrogen peroxide or a similar oxidizer. Hair straightening is a similar process, but may involve the additional use of hair dressings like gels, sprays, and creams (pomades) to hold the hair in place. Hot comb techniques may be used to disrupt the disulfide bonds followed by application of oils, petroleum jelly, or liquid paraffin .
Mousses are designed for hair styling and setting hair in position. Commonly, formulations are based on cationic conditioning polymers with quaternary ammonium salts, alcohols, perfumes, colorants, water, and preservatives. They may be in propellant, foam, or jelly form for direct application to hair after shampoo or cutting.
COMPOSITION OF TYPICAL STYLING MOUSSE
- Polyoxyethylated fatty alcohol
- Deionized water
- Polyquaternium compound chloride
- Vinyl caprolactam/polyvinylpyrolidone/dimethylaminoethyl methacrylate copolymer
- Ethanol
- Amodimethicone—tallow trimonium
- Additives (perfumes, preservatives, etc.)
SEE ALSO Fibrous Protein ; Formulation Chemistry ; Proteins .
Alan B. G. Lansdown
Bibliography
Corbett, John F. (1999). Hair Colorants: Chemistry and Toxicology. Cosmetic Science Monographs, No. 2, ed. H. Butler. Weymouth, U.K.: Micelle Press.
Gillespie, J. M. (1983). "The Structural Proteins of Hair: Isolation, Characterisation and Regulation of Biosynthesis." In Biochemistry and Physiology of Skin, ed. L. A. Goldsmith. Oxford, U.K.: Oxford University Press.
Lansdown, Alan B. G. (1995). "Action and Interaction of Metals in the Toxicology and Physiology of the Skin." CRC Critical Reviews in Toxicology 25:397–462.
Lansdown, Alan B. G. (2000). "Leads in Hair Dyes: Short Term Appeal vs. Long Term Risk." International Journal of Cosmetic Science 22:167–168.
Marzulli, F. N.; Watlington, P. M.; and Maibach, H. I. (1978). "Exploratory Skin Penetration Findings Relating to the Use of Lead Acetate in Hair Dyes." Current Problems in Dermatology 7:196–204.
Murphy, Bryan P. (2000). Hair Colorants. Poucher's Perfumes Cosmetics and Soaps, 10th edition, ed. H. Butler. Dordrecht, Netherlands: Kluwer Academic Publications.
Prota, G. (1988). "Progress in the Chemistry of Melanins and Related Metabolites." Medicinal Research Reviews 8:525–556.
Swift, J. Alan (1997). "Morphology and Histochemistry of Human Hair." In Formation and Structure of Human Hair, ed. P. Jollies, H. Zahn, and H. Höcker. Basel, Switzerland: Birkhäuser Verlag.
Swift, J. Alan (1998). Fundamentals of Human Hair Science. Cosmetic Science Monographs, No. 1, ed. H. Butler. Weymouth, U.K.: Micelle Press.