Estrogen
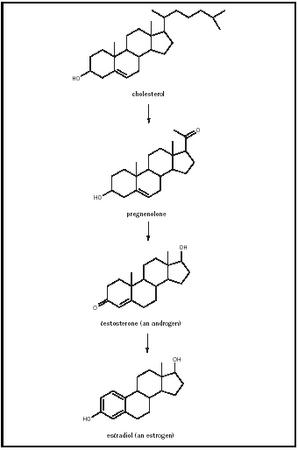
Estrogen is not one hormone, it is the name used to denote either of two steroid hormones. These hormones are noted for their role in the development of the secondary sexual characteristics of females. In mammals the most abundant (and most potent) estrogen is estradiol (a dialcohol : note the "-diol" suffix), followed by estrone (a ketone : note the - one suffix). As is true for the other steroid hormones, the major biosynthetic pathway for estrogen begins with cholesterol (C 27 ). The figure shows the major metabolic intermediates in the usual synthesis of estrogen, starting with cholesterol, proceeding to pregnenolone (C 21 ), an androgen (C 19 ), and then estrogen (C 18 ). Estrogen has an alcohol functional group at the C3 and C17 positions, and a methyl group at C13. A unique aspect of estrogen biosynthesis is the conversion (catalyzed by the enzyme aromatase) of the A ring to an aromatic ring.
Estrogen is produced in the ovaries and secreted into the blood, whereupon it enters target cells throughout the body. It enters the cells via passive diffusion and binds to specific proteins called estrogen receptors ,
which reside in the cell nuclei. In the absence of estrogen, these receptors are functionally inactive. The binding of estrogen to a receptor activates the receptor, which can then bind to specific sites on the DNA to stimulate the transcription of a highly specific set of genes. As a result, the metabolism and the character of the cell changes.
In addition to its role in the development of the secondary sex characteristics in females, estrogen has important physiological functions in the cardiovascular, immune, and central nervous systems, as well as its effects on the growth and maturation of the long bones. It is also implicated in a number of diseases (including breast and uterine cancers) and in postmenopausal health problems, such as atherosclerosis, osteoporosis, and diminution of cognitive functions. Because these conditions, especially female cancers, are associated with estrogen, a number of anticancer treatments involve estrogen blockade: drugs that prevent estrogen from binding to its receptors. As a result, the receptors remain in an inactive form.
EDWARD DOISY (1893–1986)
Before Edward Doisy earned the Nobel Prize for his research on vitamin K, he studied female hormones. It was at this time in 1936 that he isolated estradiol. In rodents without ovaries, small injections of this isolated compound were able to replicate the same behavior and biological processes as if the ovaries were still present.
—Valerie Borek
SEE ALSO Steroids .
William M. Scovell
Bibliography
Kimball, J. W. (1994). Biology. New York: W. C. Brown. Available from http://biology-pages.info .
Comment about this article, ask questions, or add new information about this topic: